Abstract
Chronic Myeloid Leukaemia (CML) is a triphasic disease which typically presents in chronic phase with risk of progression to more aggressive phases in a certain proportion of patients. Accelerated Phase (AP), as described in the pre-Tyrosine Kinase Inhibitor (TKI) era by Kantarjian et al in 1988, is an intermediate stage with a poor median overall survival (OS) of ≤18 months without haematopoietic stem cell transplantation (HSCT). Since TKI therapy has revolutionized CML treatment, a significantly improved OS has been seen in most CML patients, including those in AP. Not all CML-AP patients require HSCT upfront nowadays and many are able to achieve major molecular remission (MMR) and favourable OS on TKI therapy. However, updated classifications of CML-AP by the World Health Organization (WHO) and European LeukemiaNet (ELN) do not reflect these significant advances in the TKI era. There is a need to re-evaluate the CML-AP classification that will have an impact on treatment decisions for CML-AP patients. In this study, we explored the association between various haematological parameters at diagnosis and the probabilities of OS and progression-free survival (PFS) of CML-AP patients on TKI therapy.
Overall Survival (OS) and Progression-Free Survival (PFS) trends of 75 newly diagnosed CML-AP patients treated with frontline TKIs between 2000 to 2013 from Singapore General Hospital and Seoul St. Mary's Hospital in South Korea were retrospectively analysed with regards to demographic and haematological parameters, such as cell counts from serum and bone marrow at diagnosis, using cox proportional hazards analysis. Survival was also compared using log-rank test with Bonferroni corrections between CML-AP patients and 227 CML Chronic Phase (CML-CP) high-risk Sokal and 34 Blast Crisis (CML-BC) patients on TKI-based therapy. OS was defined as duration from diagnosis of CML-AP to death from any reason. PFS was defined as duration from disease diagnosis to the first occurrence of progression or death due to CML.
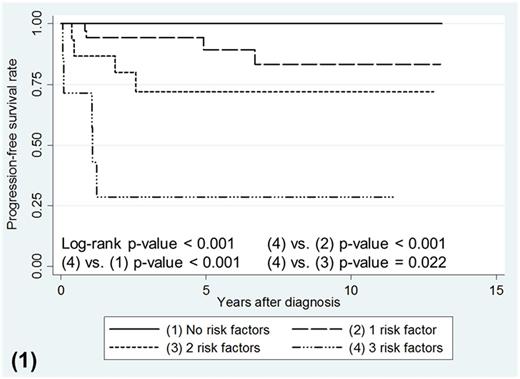
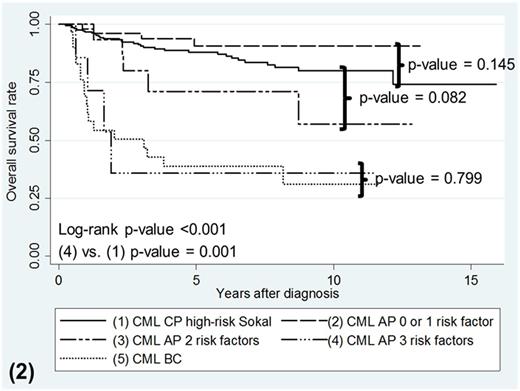
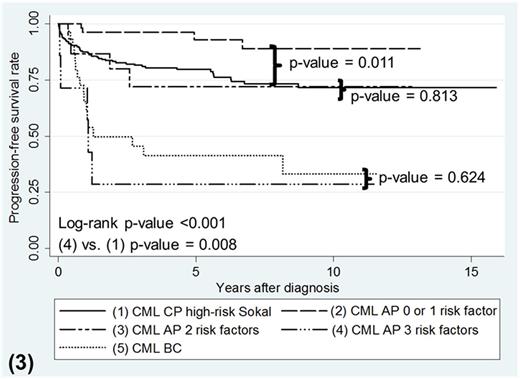
As a whole, CML-AP patients treated with frontline TKI had survival that paralleled CML-CP high-risk Sokal patients (p-value = 0.694 for OS, p-value = 0.258 for PFS). Most of the death and progression occurred less than 3 years of starting TKI therapy (69.2% for OS, 84.6% for PFS). Multivariable analysis in CML-AP patients showed that male gender, bone marrow (BM) blasts ≥10% and clonal chromosomal abnormalities (CCAs) at diagnosis were associated with poor OS (Hazard Ratio (HR) 18.53, p-value = 0.013; HR 1.16, p-value = 0.010; HR 5.05, p-value = 0.044, respectively) and poor PFS (HR 12.96, p-value = 0.021; HR 1.17, p-value = 0.007; HR 8.84.05, p-value = 0.008, respectively). CML-AP patients with all 3 of these risk factors experienced the worst OS compared to those with 1 or zero risk factors (p-value <0.001). Patients with all 3 risk factors also had the poorest PFS compared to those with 2, 1 and zero risk factors (p-value = 0.022, <0.001, <0.001 respectively; figure 1). CML-AP Patients with 2 risk factors or less, had OS and PFS probabilities comparable to CML-CP patients with high-risk Sokal score (p-value = 0.082 for OS, p-value= 0.813 for PFS, figure 2 and 3 respectively). However, CML-AP patients with all 3 risk factors showed inferior OS and PFS probabilities similar to CML-BC patients (p-value = 0.799 for OS, p-value = 0.624 for PFS; figure 2 and 3 respectively).
Our findings suggested that CML-AP was a heterogeneous group with varying survival probabilities on TKI therapy. Male gender, BM blasts ≥10% and CCAs at diagnosis were risk factors shown to be predictive of survival probabilities, and identified a high-risk sub-group among CML-AP patients with inferior OS and PFS rates similar to CML-BC patients. Aggressive chemotherapeutic strategies including HSCT should be warranted in these patients. However, TKI therapy alone with close molecular surveillance may be a reasonable option for optimally responding low-risk CML-AP patients who are not eligible for HSCT.
Kaplan-Meier survival curves for PFS according to stratification of the number of risk factors present in CML-AP patients.
Kaplan-Meier survival curves for PFS according to stratification of the number of risk factors present in CML-AP patients.
Kaplan-Meier survival curves for OS according to phases of CML with AP patients separated by number of risk factors present.
Kaplan-Meier survival curves for OS according to phases of CML with AP patients separated by number of risk factors present.
Kaplan-Meier survival curves for PFS according to phases of CML with AP patients separated by number of risk factors present.
Kaplan-Meier survival curves for PFS according to phases of CML with AP patients separated by number of risk factors present.
Kim:BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Research Funding, Speakers Bureau; ILYANG: Consultancy, Honoraria, Research Funding; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau. Goh:BMS: Honoraria; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Roche: Honoraria; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Honoraria; Takeda: Honoraria; Alexion: Honoraria, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal