Abstract
Background: Autologous stem cell transplantation (ASCT) is a standard part of first-line therapy for pts <70 years of age with multiple myeloma. If remission length is ≥ 2 years, our policy is to offer a salvage transplant upon relapse if the patient is eligible. Many centers, including ours, collect adequate CD34+ cells for two transplantations, as there is concern for poor mobilization post-therapy, including lenalidomide. However, no good data truly quantifies this rate, given plerixafor availability. The advent of novel therapies has reduced the use of second salvage ASCT (ASCT2), and the prolonged storage of cryopreserved stem cells imposes a significant economic burden. We performed a cost analysis to compare the cost of mobilizing and storing stem cells for ASCT2 in all patients versus remobilizing and collecting for ASCT2 upon relapse in eligible patients.
Methods: We developed a Markov decision-analytic model to compare the two strategies in a hypothetical cohort of 60 year old patients newly-diagnosed with multiple myeloma. The model simulates the clinical course of 10,000 patients over a 10 year time horizon, with the end point of costs per patient per strategy. Baseline probabilities were derived from published studies as well as analysis of multiple myeloma patients who underwent ASCT1 at Princess Margaret Cancer Centre between January 2003 and December 2012. Key health states include probability of progression (pPD) and death in the 1st 2 years post-ASCT1, pPD >2 years post-transplantation, probability of having an ASCT2 upon relapse vs. a non-ASCT approach. Direct costs were collected from a Canadian public health payer's perspective. Costs were obtained from hospital and provincial databases, as well as the literature and presented in 2016 Canadian dollars. Key costs collected were the cost of mobilization for 2 transplants vs. 1, the cost of remobilization and the cost of stem cell storage. Costs were discounted at 3%. All patients were assumed ASCT2 eligible at relapse. In the re-mobilization arm, all patients were successfully remobilized, with an assumed >50% rate of plerixafor use.
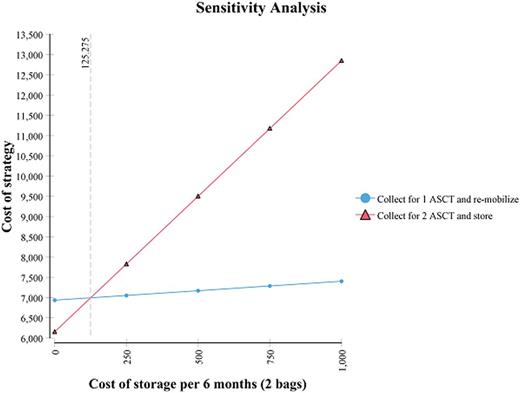
Results: 938 patients underwent ASCT1 at Princess Margaret Cancer Centre during this time period, with stem cells stored for a salvage transplant. The mean age of transplanted patients at ASCT1 was 58.4 yrs. The mean number of aphaeresis days required to collect enough cells for 2 stem cell transplants was 1.53 days. The calculated mean aphaeresis days required to collect for 1 transplant was 1.15 days. The median number of bags processed for 2 ASCT was 4, as such, after ASCT1, the median number of bags stored per person was 2. 74 patients (7.9%) underwent ASCT2 over the 10 year period. Most (73%) occurred early, 2-5 yrs post-ASCT1, with only 27% beyond this time period. Over the 10 yr horizon, the total mobilization and storage cost of the stored stem cells strategy was C$9702, versus C$7229 for the re-mobilization strategy, thus storing stem cells for a potential ASCT2 costs an extra C$2473 per patient. Our centre collects such stem cells in >100 pts/yr, at a cost of approximately $250,000. The model was robust to one-way sensitivity analyses of all variables. Storing stem cells only becomes the less costly strategy if the storage costs are less than C$125/6mo or C$10/bag/month (figure 1).
Conclusions: The use of salvage ASCT for patients who sustain at least a 2 yr remission with their first is low (less than 10%), and it is associated with significant costs to the system unless the costs of stem cell storage are minimal. With the availability of plerixafor, the cost savings would justify a re-mobilization approach for the minority of patients that are eligible for a salvage ASCT.
Prica:Janssen: Honoraria; Celgene: Honoraria. Chen:Celgene: Honoraria, Research Funding; Janssen: Honoraria, Research Funding; Takeda: Research Funding. Reece:Janssen: Consultancy, Honoraria, Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Merck: Research Funding; Novartis: Honoraria, Research Funding; Otsuka: Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; BMS: Honoraria, Research Funding; Takeda: Consultancy, Honoraria, Research Funding. Trudel:Glaxo Smith Kline: Honoraria, Research Funding; Celgene: Honoraria; Novartis: Honoraria; Oncoethix: Research Funding. Tiedemann:Takeda Oncology: Honoraria; Celgene: Honoraria; Janssen: Honoraria; Novartis: Honoraria; Amgen: Honoraria; BMS Canada: Honoraria. Kukreti:Celgene: Honoraria; Lundbeck: Honoraria; Amgen: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal