Abstract
Despite recent advances in our understanding of the genetic origins of acute myeloid leukemia (AML), clinical outcomes remain poor. While standard induction chemotherapy induces remission in most patients, the majority of patients eventually relapses and dies from progressive disease. A number of cell surface proteins have been shown to be expressed at high levels on AML stem cells compared to normal HSCs including CD47, CD44, CD96, TIM3, CD123, CD25 and IL1RAP. Despite the attention these antigens have received, data supporting their roles as cell-intrinsic regulators of LSCs are more limited. We have identified CD97, a member of the adhesion class of GPCR, as a frequently upregulated antigen expressed on AML blasts that is a critical regulator of LSC function.
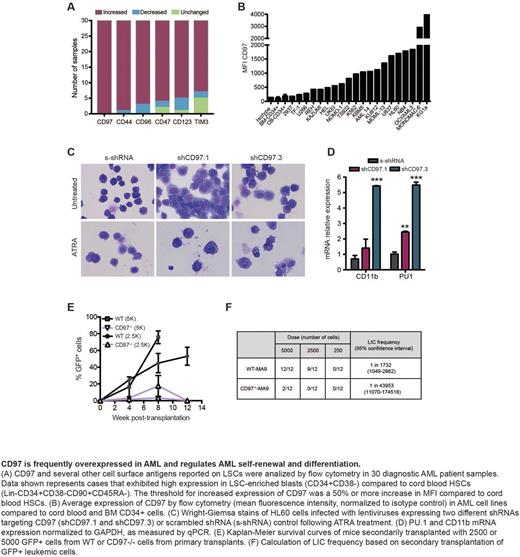
In order to determine whether CD97 is expressed on LSCs, we assessed CD97 expression in human AML primary samples by flow cytometry. This study revealed that CD97 was 10 fold increased on LSC-enriched (CD34+CD38-) blasts compared to cord blood HSCs in all 30 patients tested. In contrast, the expression of other previously reported LSC was more variable and less frequently increased over HSCs. To test if CD97 may regulate leukemic cell function, we knocked-down (KD) CD97 in human AML cell lines. CD97-KD in HL60 and MOLM-13 AML cell lines increased apoptosis, reduced cell growth 4-fold, and delayed mortality once injected into NSG mice (20 vs. 28 days, p=0.0012). As CD97 expression increases during normal myelomonocytic differentiation, we cultured control and CD97-KD HL60 or U937 cells (which can be induced to differentiate along the myelomonocytic lineage following all-trans-retinoic acid (ATRA) treatment) for 4 days in the presence of 10nM ATRA or vehicle control. Flow cytometric analysis revealed that CD97-KD cells expressed higher levels of the myeloid differentiation antigens CD11b and CD15 both before and after ATRA treatment. In addition, mRNA expression of CD11b and PU.1, key genes associated with myeloid differentiation, were increased in CD97-KD cells compared to controls.
To investigate whether CD97 regulates leukemic initiation, purified HSPCs from WT and CD97-/- mice were transduced with the MLL-AF9 oncogene (MA9). CD97-/--MA9 cells (GFP+) showed a 3-fold reduction in colony formation in serial replating assays in methylcellulose, consistent with a reduction in self-renewal. Mice transplanted with 5,000 CD97-/--MA9 blasts survived significantly longer than those receiving WT-MA9 blasts (70 vs 100 days, p=0.0039). Consistent with reduced disease aggressiveness, at the time of sacrifice mice injected with WT-MA9 cells contained a higher percentage of total GFP+ cells, L-GMPs and CD11b+Gr1+ cells in the BM than those receiving CD97-/--MA9 grafts. Secondary transplant experiments revealed that CD97-/--MA9 blasts exhibit a 25-fold lower LIC frequency than WT-MA9 blasts. Similar results were obtained using the AML1-ETO9a mice model.
To better understand the molecular pathways underlying CD97 regulation of AML cells, we performed a combination of RNA-sequencing and protein phospho-array analyses in CD97-KD and scramble control cells. Gene Ontology and GSEA analyses revealed enrichment of transcripts encoding genes that regulate apoptosis as well as downregulation of cell cycle regulators in CD97-KD cells. Consistent with our flow cytometry data, CD97-KD induced differentiation, as evidenced by increased expression of myeloid differentiation genes and global increases in granulocyte and monocyte fingerprint signatures. Phosphoproteome analysis revealed downregulation of several mediators of the MAPK cascade and MTOR signaling pathways in CD97-KD cells. These signaling changes were confirmed by western blot, as activating phosphorylation events of ERK 1/2, c-Jun, and STAT3 were decreased upon CD97-KD. Together, these data indicate that functional alterations in CD97-KD AML cells are due to the modulation of signaling cascades including MAPK and mTOR.
Our studies indicate that CD97 is expressed at a higher frequency in AML patient samples than previously identified LSC antigens and that its expression is elevated, irrespective of genetic subtype. Studies using human cell lines and mouse models of AML demonstrate that CD97 regulates the proliferation and survival of leukemic blasts as well as disease initiation and self-renewal. Collectively, these data credential CD97 as a promising therapeutic target in AML.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal