Abstract
Introduction: Multiple myeloma (MM) is characterized by malignant plasma cell (PC) proliferation. Gene expression profiling (GEP) of CD138+ bone marrow PC has emerged as a new way to identify patients with worse clinical outcomes so that an individualized treatment approach can be undertaken in the clinic. The majority of data with GEP come from clinical trials. We aimed to evaluate the use of GEP in a standard clinical setting.
Methods: We retrospectively searched our database of newly diagnosed MM patients with GEP completed prior to initial treatment. 35MM patients from April 2014 until June 2015 were identified and included in our analysis. GEP was performed through MyPRS® (Signal Genetics, Little Rock, AR). Fisher's exact test was used to evaluate the associations between complete response status and other categorical variables. The Wilcoxon rank sum test was used to evaluate the difference in continuous variables between patients that achieved a complete remission after 6 months of initial treatment and those who did not. Responses were assessed using IMWG criteria.
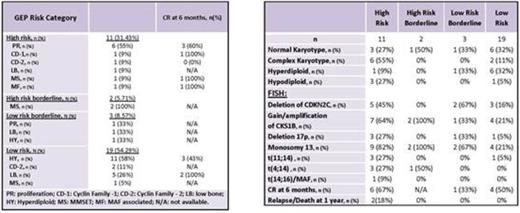
Results: Median age was 60 (38-76). Patients presented with lytic lesions (60%), anemia (80%), kidney dysfunction (11%) and hypercalcemia (20%). All patients with known initial therapy were treated with bortezomib (n=29) or carfilzomib (n=1) based therapy. 10 patients had upfront autologous stem cell transplant. 18 patients had available response at 6 months. 37% (n=13) of patients were characterized as high risk by GEP, of which 46% (n=6) had the proliferation (PR) subtype. Most low risk patients had hyperdiploidy (HY) subtype (n=12, 55%). Patients with high risk GEP presented more often with complex karyotypes whereas low risk GEP patients most often had normal or hyperdiploid karyotypes. FISH abnormalities that are usually present in high risk myeloma were also present in patients classified as GEP low risk (Table 1). At 6 months after diagnosis, lower baseline total protein, serum M-spike, serum free light chain ratio and serum kappa light chain levels were significantly associated with achieving a stringent complete response (sCR) (p<0.05). High risk patients were more likely to achieve a CR (n= 6/10, 60%) than low risk patients (n=5/16, 31%) at 6 months. Despite this, high risk patients seemed more likely to lose CR and relapse or die. Within one year of diagnosis, one patient relapsed after achieving a VGPR (high risk, PR subtype) and one patient died due to MM progressive disease after achieving an sCR (high risk, CD-1 subtype) (Table 1).
Conclusion: In a standard clinical setting, GEP seems to identify MM patients that are at higher risk of adverse clinical outcomes early after diagnosis. GEP may be an adjunct to cytogenetics/FISH in MM risk stratification, as high risk FISH abnormalities were also found in low risk GEP patients. Larger studies with longer follow up may help address the particular role of GEP in the individualized treatment of MM.
Orlowski:Array BioPharma: Consultancy, Research Funding; Acetylon: Membership on an entity's Board of Directors or advisory committees; Onyx Pharmaceuticals: Consultancy, Research Funding; Spectrum Pharmaceuticals: Research Funding; Celgene: Consultancy, Research Funding; Genentech: Consultancy; Millennium Pharmaceuticals: Consultancy, Research Funding; Forma Therapeutics: Consultancy; BioTheryX, Inc.: Membership on an entity's Board of Directors or advisory committees; Janssen Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal