Abstract

Background
Iron overloading is a common problem for adult with myelodysplastic syndrome (MDS), aplastic anemia (AA) or other chronic anemia. Deferasirox (DFX) has been proven as an effective therapy to chelating iron in these patients. Anyway, the safety of DFX is still a concern, and the information of safety profiles and efficacy are less understood in Taiwan. This study is planned primarily to collect long-term safety data of DFX treatment in iron-overloaded MDS, AA and other chronic anemia patients in Taiwan.
Study Design
This is an observational, single-arm, and multi-center study. Low-risk MDS or AA patients with transfusion-related iron overload, or patients with other anemia and serum ferritin more than 2000 ug/ml, were enrolled within the 18-month enrolling period if DFX is planned to be prescribed. Exposure of iron chelating agents other than DFX before trial initiation was allowed. The initial dose and subsequent adjustment of DFX were up to investigator¡¦s preference. All patients were followed for 3 years for adverse events (AEs) and disease outcomes.
Results
From 2009 to 2011, 79 patients were enrolled in this study, including 38 MDS, 23 AA and 18 other chronic anemias. Forty-seven cases (59.5%) were male, with mean age 64.3¡Ó17.8 y/o. Fifty-six (70.9%) subjects failed to complete the 3-year study period, but only 8 (10.1%) of the subjects withdrew DFX due to drug-related AEs. The mean DFX exposure dose during study was 17.7¡Ó4.02 mg/Kg/day. In contrast with those reported in literature, the most frequently reported drug-related AEs were rash (16, 20.3%), diarrhea (11, 14.0%), hypercreatinemia (8, 10.1%), pruritus (7, 8.9%), and so on (as in Table 1). When classified by organ systems, skin disorders were the frequently reported one (26, 32.9%), and followed by GI disorders (n=24, 30.4%). Grade 3-4 drug-related adverse events were rare (n=4, 5.1%).
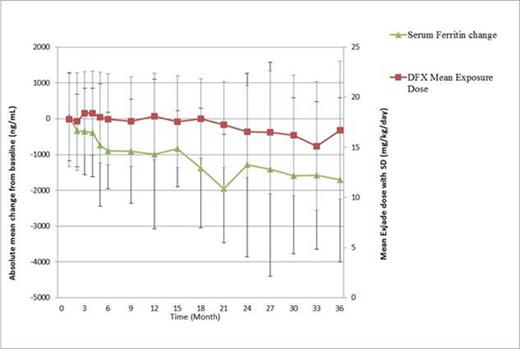
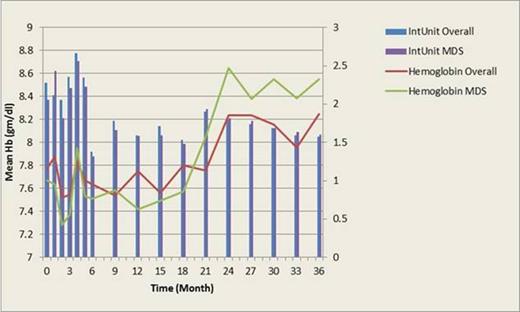
For all subjects, DFX could effectively decrease serum ferritin level from baseline (-985+/-2090 ng/ml (p=0.0154 vs. baseline) and -1710+/-2290 ng/ml (p=0.0424 vs. baseline) at 1 yr and 3 yr, respectively) (as in Figure 1). Notably, after DFX usage, 23 patients (32.4%) developed erythroid response according to IWG 2006 criteria; the mean hemoglobin could increase from 7.77+/-1.63 gm/dl (baseline) to 8.25+/-2.60 gm/dl (at 36 month, p=0.6172 vs. baseline), when the average transfusion amount was decreased from 2.3+/-1.4 units (baseline) to 1.6¡Ó0.5 units (at 36 months, p=0.0406 vs. baseline). (as in Figure 2). Ten patients (10/46, 21.7%) had platelet response.
For the 38 MDS patients, DFX also could significantly lower serum ferritin level (-590+/-2490 ng/ml (p=0.4095 vs. baseline) and -1310+/-362 ng/ml (p=0.0013 vs. baseline) at 1 yr and 3 yr, respectively) but seemed to have a less extent than that in overall population. Similarly, 10 patients (21.7%) developed erythroid response after DFX use. The mean hemoglobin increment (from 7.67+/-1.67 gm/dl (baseline) to 8.55+/-3.45 gm/dl (at 36 month, p=0.6012 vs. baseline)) and the decrease of average transfusion amount (from 2.1+/-1.2 units (baseline) to 1.6+/-0.6 units (at 36 months, p=0.2943 vs. baseline) were not significant, probably due to low case number (as in Figure 2). Four (4/19, 21.1%) patients experienced platelet response.
Conclusion
This study showed that the profiles of AEs regarding DFX use for adult anemic patients with transfusion-related iron overload in Taiwan were significantly different from those reported in Western countries. The AE-related discontinuation rate was also relatively low. An expected efficacy to lowering serum ferritin by DFX, and a significantly degree of hematological improvement was noted, too.
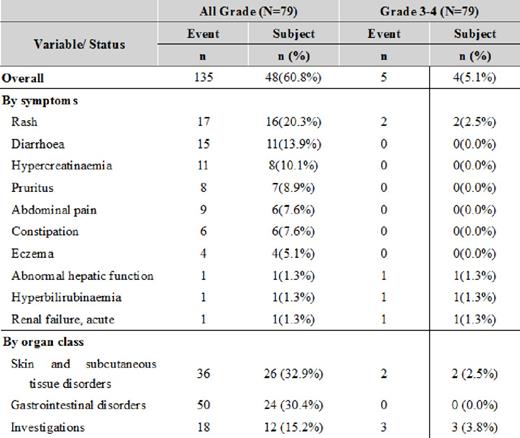
Drug-related adverse events, for all events with incidences > 5% and all grade 3-4 events:
Drug-related adverse events, for all events with incidences > 5% and all grade 3-4 events:
Chang:Novartis: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal