Abstract
Background: In utero hematopoietic cell transplantation (IUHCT) is a nonmyeloablative, nonimmunosuppressive transplant approach that results in donor cell engraftment across immune barriers. Although a significant amount of work has investigated the fate of T cells following IUHCT, little attention has been paid to B cell tolerance and the fate of donor derived host reactive or host derived donor reactive B cells following IUHCT. B cell tolerance is broadly believed to occur by a combination of 3 mechanisms: deletion, receptor editing, and functional inactivation (anergy). In the current study we attempt to elucidate the mechanism(s) by which B cell tolerance occurs following IUHCT.
Methods: 10x106 donor bone marrow (BM) cells were injected intravenously via the vitelline vein into gestational day 14 murine fetuses. IUHCT was performed in the congenic (C57Bl/6-GFP [H2Kb ] into C57Bl/6 [H2Kb ]) and allogeneic (C57Bl/6-GFP into Balb/c [H2Kd ]) strain combinations. Naive Balb/c and C57Bl/6 mice served as controls. Mice were sacrificed at day of life 3 (P3), 1 month and 4 months of age at which time their BM, spleen, and serum were harvested. To assess B cell deletion, flow cytometry was used to determine the absolute # and % of host and donor immature and pre B cells in the BM. Additionally, apoptosis of host and donor BM derived B cells was determined by annexin staining. Central receptor editing was evaluated using RT qPCR to measure the amount of Vκ-RS rearrangments in BM pre-B cells. Peripheral receptor editing was studied by calculating the % of λ light chains in mature splenocytes identified by flow cytometry. Finally, functional inactivation of donor reactive host B cells was assessed by measuring anti-H2Kb serum antibodies (ab) of allogeneic chimeras, naive, and immunized mice at 1 month of age.
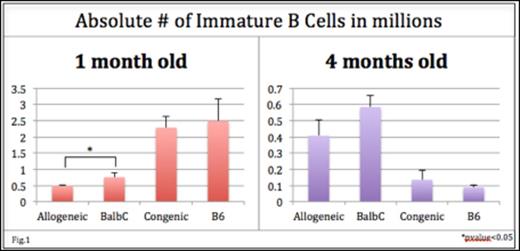
Results: The absolute number of BM immature B cells was decreased in allogeneic recipients of IUHCT compared to noninjected Balb/c controls at 1 month of age (fig 1). This effect was lost by 4 months of age. The decrease in B cells resulted primarily from a decrease in immature donor as opposed to host B cells compared to controls (% immature donor B cells in allogeneic recipients vs. controls: 16.2% vs. 39.9%; p<0.0005). Donor B cells in allogeneic chimeras also demonstrated a trend toward increased apoptosis compared to controls (24.8 vs. 18.7%; p=0.2) which was not seen in immature host B cells (18.3 vs. 18.6%; p=0.9). There was no significant decrease in the absolute number of immature B cells or increased apoptosis in congenic recipients compared to uninjected controls. These findings suggest deletion of autoreactivedonor B cells.
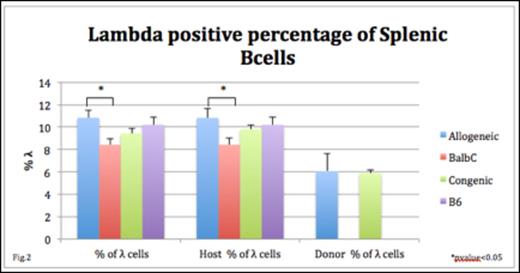
Light chain receptor editing involves rearrangements within the κ and λ gene loci and may occur in BM pre-B cells or mature B cells in the spleen. We found no difference in the Vκ-RS rearrangements of pre B cells in allogeneic chimeras and controls at 1 month. In contrast, the quantity of total λ+ mature splenic B cells was increased in allogeneic chimeras at P3 (10.8 vs. 8.4%; p=0.02) and resulted from an increased host λ+ % compared to controls (10.8 vs. 8.4% p=0.03) suggesting peripheral receptor editing of host cells (fig.2). The λ+ % increase in allogeneic chimeras was lost by 1 month.
Autoreactive B cells that escape deletion and receptor editing can be functionally inactivated. Neither allogeneic nor naive mice developed ab to H2Kb splenocytes, however, Balb/c mice immunized to H2kb antigen showed high ablevels (MFI fold change: allo-0.89 naive-1.37 imm-2.77; p<0.05).
Conclusion:
B cell tolerance after IUHCT is achieved by distinct mechanisms for host and donor cell populations. Donor derived host reactive B cells undergo deletion and apoptosis while receptor editing and functional inactivation are the primary mechanisms of B cell tolerance of host derived donor reactive B cells. We hope use this and future studies of antigen specific B cell tolerance to harness the immunologic potential of IUHCT for many hematopoietic and immunologic congenital diseases.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal