Abstract
Background and Methods: Lenalidomide is an immunomodulatory agent with promising activity in CLL, including various stimulatory effects on T cells (Riches & Gribben, Semin Hematol 2014). This phase II study was conducted in advanced-phase CLL patients and explored the in vivo effects of low-dose lenalidomide on T cell proliferation and immune checkpoint molecule expression. Lenalidomide was used as a single agent week 1-4, after which alemtuzumab was added week 5-16 (as a strong T-cell depleting agent). The safety and clinical efficacy of the combination was also analyzed. Immune monitoring was performed at baseline, after 4 and 16 weeks (end of treatment, EoT) as well as during long-term follow-up. T cells were analyzed by flow cytometry for the cell proliferation marker Ki67, the activation marker HLA-DR and the immune checkpoint markers PD-1 and CTLA-4. CLL cells were analyzed for PD-1 ligand (PD-L1) expression.
Results: Totally 23 patients were included. The median age was 69 years (range 61-85), 70% (16/23) had Rai stage III/IV, the median number of prior regimens was 4 (range 1-7) and 61% (14/23) had 17p and/or 11q deletion. The overall response rate (IWCLL criteria) was 58% (11 of 19 evaluable patients), including two CR and nine PR. Median progression-free survival was 5 months (range 0-37+). Median response duration was 11 months (range 1-29+ months). Grade III-IV neutropenia or thrombocytopenia occurred in 84% and 55% of patients, respectively. The most common non-hematological grade III-IV adverse event was febrile neutropenia (7/23 patients, 30%). CMV reactivation requiring valganciclovir therapy occurred in seven patients (30%). The maximum tolerated dose of lenalidomide was 5 mg/day.
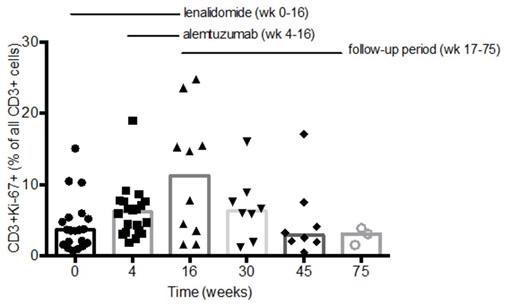
A significant increase in the proportion of proliferating T cells (CD3+/Ki67+) was observed after single agent lenalidomide treatment, from a median of 3.6 % (range 0.8-15) at baseline to 6.2 % (range 1.9-19) at week 4 (p=0.003). The proportion of Ki67+ T cells increased further after adding alemtuzumab with the peak value observed at EoT, after which normalization occurred gradually during follow-up (Figure 1). Even though the total number of T cells was low at EoT due to alemtuzumab, the percentage of Ki67+ T cells in both CD4+ and CD8+ cell subsets had increased significantly (median 6.2% and 9.5 % respectively at week 16, p=0.01 and p=0.02). Furthermore we observed a significant increase in the proportion of HLA-DR positive T cells during therapy (p=0.001 in the CD4+ subset and p=0.02 in the CD8+ subset). The Th1/Th2 balance did not change after four weeks of lenalidomide treatment but a significant increase in Th2 cells was observed after combination treatment (p=0.02). The median baseline PD-L1-expression on CLL cells was 0.3% (range 0.0-0.7). A median of 25.2 % (range 9.3-42.4) of CD4+ T cells and 6.2 % (range 2.6-21.7) of CD8+ T cells were PD-1 positive. The median baseline expression of CTLA-4 was 0.01% (range 0.0-0.5) in CD8+ T cells and 0.1 % (range 0.01-1.13) in CD4+ T cells respectively. However, no significant changes in PD-1, PD-L1 or CTLA-4 expression were observed, neither after 4 weeks of lenalidomide single agent therapy nor during combination treatment with alemtuzumab.
Conclusions: A significant increase in the proportion of Ki67-positive T cells was observed during low-dose lenalidomide treatment that was not eliminated by depleting overall T cell numbers in vivo by alemtuzumab. Immune checkpoint molecule expression remained largely unaffected. Lenalidomide and alemtuzumab in combination showed clinical activity and an acceptable safety profile in patients with advanced, heavily pretreated CLL. Further studies are warranted on the complex role of T cells in CLL, their responsiveness to lenalidomide as well as drug induced immune-enhancing effects in other clinical situations.
Winqvist:Janssen Cilag: Research Funding. Off Label Use: Lenalidomide is not approved for CLL.. Mellstedt:Celgene: Research Funding. Osterborg:Gilead: Honoraria; Janssen: Honoraria, Research Funding; GSK: Research Funding; Pharmacyclics LLC, an AbbVie Company: Research Funding; Amgen: Research Funding. Lundin:Janssen: Research Funding; Novartis: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal