Abstract
Introduction. The standard first-line treatment of young and fit mantle cell lymphoma (MCL) patients consists of a rituximab containing induction of CHOP and high dose ARA-C, followed by high dose consolidation and autologous stem cell transplant (ASCT). Thus far, almost all patients relapse after ASCT. Bortezomib is a proteasome inhibitor with activity in MCL. We investigated in a randomized phase II study whether there was any indication that maintenance therapy with bortezomib after ASCT could improve the outcome of treatment, measured as event free survival (EFS).
Methods. Patients 18-65 years with newly diagnosed MCL were treated with 3 cycles of R-CHOP and 2 cycles of ARA-C (2 x 2 g/m2 iv d1-4) and rituximab (375 mg/m2, iv d11). Patients in PR or CR continued with ASCT after BEAM conditioning. Patients with a PR or CR after ASCT, with a neutrophil count > 0.5 x 109/l and platelets > 80 x 109/l were randomized between bortezomib and no further treatment. Bortezomib 1.3 mg/m2 iv was given once every two weeks, for 2 years, starting between 6 - 12 weeks after transplantation.
Results. Between October 2007 and February 2012, 140 patients aged 34-66 years (median 57) were registered. Five patients were not eligible. The MIPI score was low, intermediate, high or unknown in 57%, 32%, 10% and 1% respectively. All eligible patients started induction treatment with R-CHOP. Two patients did not receive the first ARA-C cycle, because of progressive disease and physician decision. Hundred-fifteen patients (85%) received the BEAM and ASCT. Reasons to stop protocol treatment before ASCT were: progressive disease (PD) (n=6), inadequate stem cell harvest (n=3), excessive toxicity (n=3), other reasons (n=8). The response after ASCT was CR/CRu in 99 patients (86%), PR in 15 (13%), unknown in 1 (1%).
Only 62 patients (45%), aged 34-65 years (median 56) were randomized between bortezomib maintenance and no further treatment. Two patients were randomized, but not eligible. Reasons for no randomization were: not eligible (n=30), patient refusal (n=14), excessive toxicity (n=2, and other reasons (n=7). In each treatment arm 30 patients were included. The patient characteristics were well balanced, except the MIPI score. In the no maintenance arm 21 patients (70%) had a low MIPI, compared to 15 patients (50%) in the bortezomib arm, and 6 (20%) versus 11 (37%) patients had an intermediate MIPI score. In both arms 3 patients (10%) had a high MIPI. Fifteen patients (50%) in the maintenance group continued bortezomib for 2 years. Reasons to stop earlier were excessive toxicity (n=6), PD/relapse (n=4), patients refusal (n=3) and other (n=2).
The main adverse events during maintenance therapy were neurologic (CTC grade 2: 14%, grade 3: 4%), and infectious (grade 2: 11%, grade 3: 7%). Three patients developed a secondary malignancy: a basal cell carcinoma in the no maintenance group, a melanoma and a prostate carcinoma in the bortezomib group.
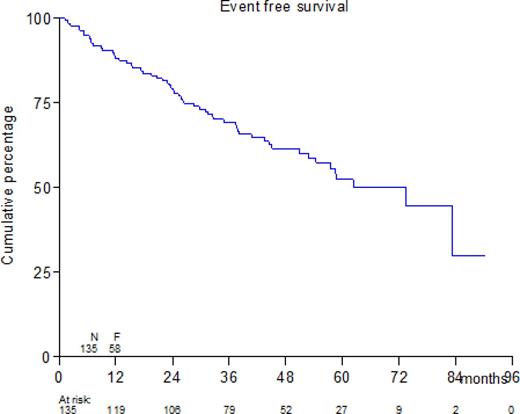
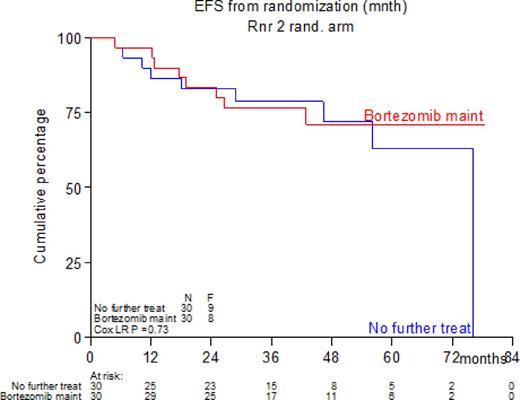
With a median follow-up of the patients still alive of 50.9 months EFS at 4 years for all patients is 61%, and the overall survival (OS) 78%. The median follow-up of the randomized patients still alive is 42.9 months. The EFS at 4 years is 72% without maintenance versus 71% in the patients randomized for bortezomib maintenance. The OS at 4 years also shows no significant difference between the groups, 90% versus 93% respectively.
Conclusion. There is no indication that bortezomib iv maintenance in a frequency of once every 2 weeks does improve the EFS of newly diagnosed MCL patients after intensive induction treatment with R-CHOP, double ARA-C and BEAM followed by ASCT.
Doorduijn:Celgene: Consultancy; Janssen: Consultancy; Roche: Consultancy. Off Label Use: bortezomib maintenance in MCL. Minnema:Celgene: Consultancy; Jansen Cilag: Consultancy; Amgen: Consultancy. Kersten:janssen: Honoraria, Research Funding; takeda millennium: Research Funding; roche: Honoraria, Research Funding. Lugtenburg:Mundipharma: Consultancy; Servier: Consultancy; Janssen-Cilag: Consultancy; Celgene: Consultancy; Roche: Consultancy. Schipperus:Novartis: Consultancy. Zijlstra:Celgene: Consultancy; Roche: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal