Abstract
Background: The addition of R has been shown to improve results for pts with relapsed DLBCL who undergo BEAM (carmustine, etoposide, cytarabine, melphalan) high-dose chemotherapy followed by an autologous stem cell transplantation (ASCT) (Khouri IF, J Clin Oncol 2005;23;2240). The incorporation of radiolabeled antibodies such as yttrium-90 ibritumomab tiuxetan to conditioning regimens has also been evaluated without additional toxicity (Khouri IF, ASH 2007, abstract 620). Subsequently, a randomized trial which was undertaken at our center to compare these 2 regimens was closed early because of slow accrual. Herein, we compare the long-term outcome in pts treated in these trials.
Methods: A combined analysis was carried out from 113 pts. Between 1999 and 2003, 57 pts with relapsed DLBCL were enrolled on a protocol with BEAM conditioning plus R at 1000 mg/m2 on days +1 and +8 after ASCT (Group A). Between 2004 and 2006, a similar group of 26 patients were entered onto a trial consisting of ibritumomab tiuxetan plus BEAM (Z/BEAM). Ibritumomab tiuxetan was given at the fixed doseof 0.4 mCi/Kg on day -21 followed by BEAM(days -7 to -1) (Group B). In 2007-2010, a randomized trial was undertaken comparing BEAM/R (Group C, n=16) and Z/BEAM (Group D, n=14). All pts received R during stem cell collection, administered at 375 mg/m2 on the day before initiating chemotherapy for stem cell mobilization, and again at 1000 mg/m2, 7 days later. The same eligibility criteria were used for all groups. In addition, pts who were enrolled on the randomized trial (Groups C and D) underwent FISH analysis for -5 and -7 and cytogenetic analysis by G-banding pre-enrollment; those who had a clonal abnormality were excluded. We also retrospectively evaluated the histologic subtypes of mediastinal, transformed and de novo DLBCL. We determined the cell-of-origin of the latter using the Visco/Young and Choi W, et al. immunohistochemical algorithms. A univariate analysis was conducted for factors of interest: this included the group conditioning, age, sex, number of prior treatments, disease status at transplantation (CR/PR), IPI (0 vs >0), LDH, β2-microglobulin, PET status, and histology subtype. Multivariate survival analysis was then conducted using backward elimination on the basis of the likelihood ratio test and including the conditioning regimens and all the factors with P < 0.05 in the univariate analyses.
Patients: There was no significant difference in the prognostic factors described above between the 4 groups with the exception of prior treatments. More pts in the BEAM/R groups received > 2 prior chemotherapies than the Z/BEAM groups (32%, 11.5%, 69% and 50% in Groups A, B, C and D, respectively; P < 0.001). Non-GCB histology within the 4 groups was present in 39%, 26%, 31% and 21%, respectively (P=0.31).
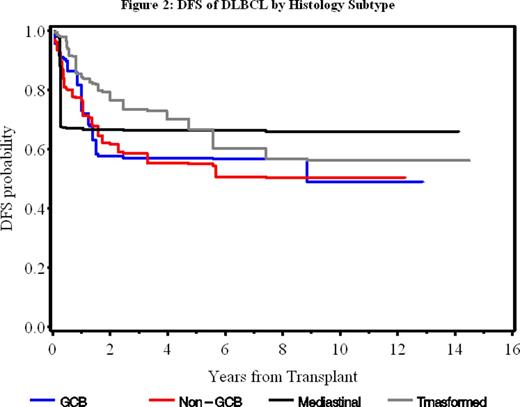
Results: The median follow-ups times for Groups A, B, C and D were 11.8, 8.1, 4.2 and 4.9 years, respectively. The 5-year overall survival (OS) rates for these groups were 74%, 73%, 69% and 86%, respectively (P = 0.46) and the disease-free survival (DFS) rates were 62%, 65%, 63%, and 63%, respectively (P = 0.99) (Figure 1). Multivariate analysis at 5-year showed that previous exposure to >3 prior chemotherapies and IPI were predictors for OS (P= 0.003 and 0.005, respectively), and DFS (P = 0.003 and 0.006, respectively). There was no significant difference in OS or DFS rates between GCB, non-GCB, mediastinal and transformed DLBCL (Figure 2). The 5-year non-relapse mortality for all pts was 4.7% and the 5-year relapse rate was 32.5% (34.0%, 30.8%. 25% and 37.5% for Groups A, B, C and D, respectively). The 5-year risk of secondary myelodysplasia (MDS) for all pts was 6.2%. None of the pts in Groups C and D who had pre-transplant cytogenetic testing developed MDS at 5-year.
Conclusions: Long-term follow-up results show no difference in OS, DFS, relapse rate, or risk of MDS between Z/BEAM and BEAM/R after in-vivo purging with R during stem cell collection. Our results were reproducible in 3 trials and appear to be superior to published reports in the literature of similar trials without in-vivo purging. This highlights the importance of this procedure for ASCT in DLBCL.
Off Label Use: The use of Zevalin and rituximab in transplantation. Alousi:Therakos, Inc: Research Funding. Fanale:Merck: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Research Funding; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Honoraria, Research Funding; Infinity: Membership on an entity's Board of Directors or advisory committees; Spectrum: Membership on an entity's Board of Directors or advisory committees; Seattle Genetics: Honoraria, Research Funding; Genentech: Research Funding; Medimmune: Research Funding; Novartis: Research Funding; Bayer: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees; Molecular Templates: Research Funding; ADC Therapeutics: Research Funding; Onyx: Research Funding; Gilead: Research Funding. Nastoupil:AbbVie: Research Funding; Janssen: Research Funding; Celgene: Honoraria; TG Therapeutics: Research Funding; Genentech: Honoraria. Westin:Spectrum: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal