Abstract
BACKGROUND: Peripheral T-cell lymphomas (PTCL) encompass a heterogeneous group of neoplasms accounting for 10 to 15% of non-Hodgkin lymphomas worldwide. Prognosis for PTCL patients is poor and consolidation in first remission with autologous stem cell transplantation (ASCT) is widely used. Most patients though still relapse after transplant. We hypothesized that pre-ASCT bone marrow (BM) involvement detected by multi-parameter flow cytometry (FC) would identify patients with inferior outcome after ASCT.
METHODS: We retrospectively analyzed the outcome of 29 consecutive PTCL patients who underwent ASCT at the Fred Hutchinson Cancer Research Center from April 2004 through July 2014. Pre-ASCT BM involvement by flow cytometry (FC) was defined as the presence of an abnormal T-cell population detected by multi-parameter FC analysis in a BM aspirate obtained within 30 days prior to ASCT. An abnormal T-cell population accounting for a percentage equal or greater than 0.01% of total leukocytes after red blood cell lysis was considered significant.
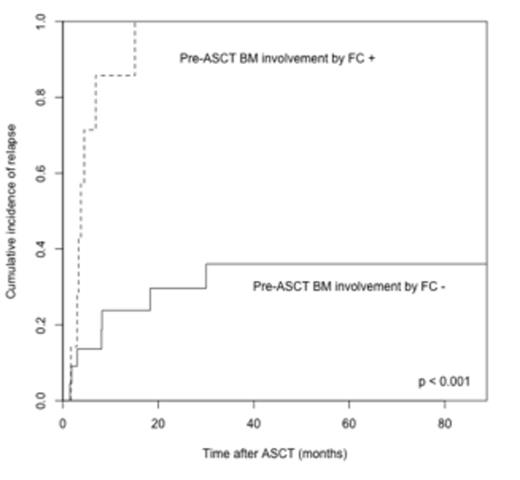
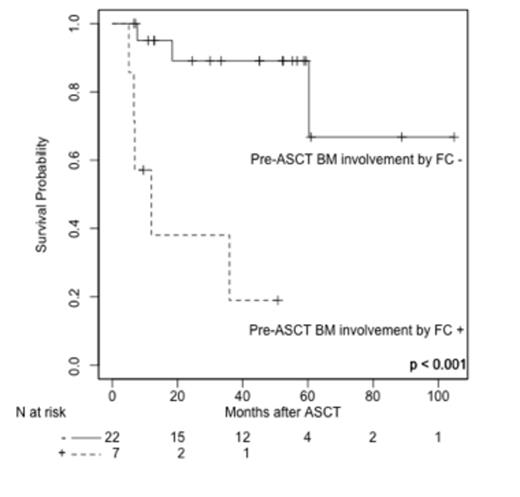
RESULTS: Ten patients (34%) with angioimmunoblastic T cell lymphoma (AITCL), 8 (27%) with ALK-negative anaplastic large cell lymphoma (ALCL), 8 (27%) with peripheral T cell lymphoma, not otherwise specified (PTCL-NOS) and 3 (12%) with other PTCL sub-types were included. Median age at transplant was 54 (range: 29-71). Twenty patients (76%) presented at ASCT in complete remission (CR) per 1999 Cheson criteria and 9 (31%) were in first CR (CR1). Fifteen patients (50%) underwent ASCT upfront. Pre-ASCT BM involvement was detected by FC analysis in 7 patients (24%, 3 patients with AITCL, 1 patient with ALCL and 3 patients with PTCL-NOS) and by morphology in 2 patients (7%, 2 patients with AITCL). The 7 patients with pre-ASCT BM involvement detected by FC experienced a significantly higher 4-year cumulative incidence of relapse (CIR) (85% versus 36%, p < 0.001) and lower overall survival (OS) (19% versus 89%, p < 0.001) with a median follow-up of 51 months. These findings were confirmed in multivariable analysis for CIR (HR = 7.37, CI = 1.14 - 47.61, p = 0.03) and OS (HR = 7.04, CI = 1.29 - 38, p = 0.024). The IPI score, absence or presence of CR1 and the number of prior therapies were included in a multivariable model for CIR, while age and the IPI score at diagnosis were taken into account for OS. ASCT performed in CR1 was associated with lower CIR (HR = 0.11, CI = 0.03-0.041, p < 0.001). Histologic subtypes did not impact CIR (p = 0.97) nor OS (p = 0.91) and they did not significantly differ between the groups with and without pre-ASCT BM involvement detected by FC (p = 0.60) after applying Fisher's exact test. Further analysis of the 22 patients presenting in CR at ASCT revealed that in this subgroup, pre-ASCT BM involvement by FC (n = 4) was also associated with higher CIR (75% versus 32%, p = 0.002) and lower OS (25 versus 94%, p <0.001). The prognostic impact of pre-ASCT BM involvement by FC persisted in multivariable analysis for CIR (HR = 16, CI = 1.11-228.70, p = 0.042, IPI score at diagnosis and number of prior therapies considered as covariables) but not for OS (HR = 0.15, CI = 0.65-65.40, p = 0.10, multivariable model including age and the IPI score at diagnosis).
CONCLUSION: Pre-ASCT BM involvement by FC correlated with dramatically higher relapse rates and an inferior OS in PTCL patients after ASCT. Furthermore, we demonstrate that residual disease, detected only in patients meeting Cheson 1999 criteria for CR at ASCT is capable of predicting a higher risk for relapse. These findings should encourage further evaluation of minimal residual disease in PTCL patients achieving complete remission as defined per Cheson 1999 and 2007 criteria.
Gopal:Merck: Research Funding; Emergent/Abbott: Research Funding; Cephalon/Teva: Research Funding; BioMarin: Research Funding; Sanofi-Aventis: Honoraria; Millenium: Honoraria, Research Funding; Seattle Genetics: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Spectrum: Consultancy, Research Funding; Gilead: Consultancy, Research Funding; Piramal: Research Funding; Biogen Idec, BMS: Research Funding. Maloney:Juno Therapeutics: Research Funding; Roche/Genentech: Honoraria; Janssen Scientific Affairs: Honoraria; Seattle Genetics: Honoraria. Till:Roche-Genentech: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal