Abstract

Objective:
Relapse of high risk hematologic malignancies remains the major cause of mortality after matched sibling and matched unrelated allogeneic hematopoietic stem cell transplantation (HSCT). A second transplantation using the same donor or another fully matched donor yielded similar results with no clear advantage of choosing a newly matched donor (1). The purpose of this analysis was to evaluate the efficacy of HIDT with pTcy as a second HSCT among patients with high risk hematologic malignancies relapsing after a matched sibling or a matched unrelated donor.
Methods and Population:
All consecutive patients (n=20) who underwent a HIDT using ptCy at our center as a second allogeneic transplant for relapse of malignancy following a prior HLA-matched transplant were included in this retrospective analysis. Patient, disease and transplant related data was obtained from our institutional BMT database where it had been prospectively documented. Survival and disease-free survival (DFS) were estimated using the Kaplan-Meier method, Relapse and non-relapse mortality (NRM) were treated as competing risks. GVHD was prospectively documented and graded.
Results:
Patients (male n=13, female n=7) had a median age of 54 years (range 21-64). The median time from the first to the second HSCT was 20.7 months (range 2.7-65.8 months). 10 patients had AML/MDS, 6 ALL, 2 CLL and 2 myeloproliferative syndrome. Grafts from the first HSCT were 50% matched related (n=10) and 50% matched unrelated (n=10). The median number for HLA mismatches among HIDT recipients was 5/10 (range 4/10-8/10). All patients received cytoreductive therapy prior to the HIDT with 12 (60%) achieving a CR and 8 (40%) with active disease at the time of conditioning regimen initiation for HIDT. The conditioning regimen for HIDT was myeloablative in 3 patients (15%) with fludarabine/high dose TBI/pTcy in 2 (10%) patients and flu/busulfan/Cytoxan in 1 patient (5%) and non-ablative/reduced intensity (flu/low dose TBI/Cytoxan n=17; in 18 (90%) patients. All patients received pTcy and tacrolimus plus mycophenolate for graft versus host disease prophylaxis.
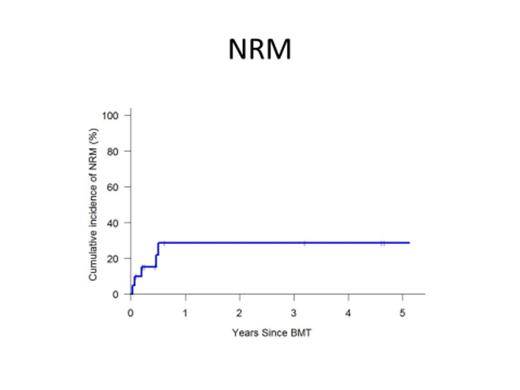
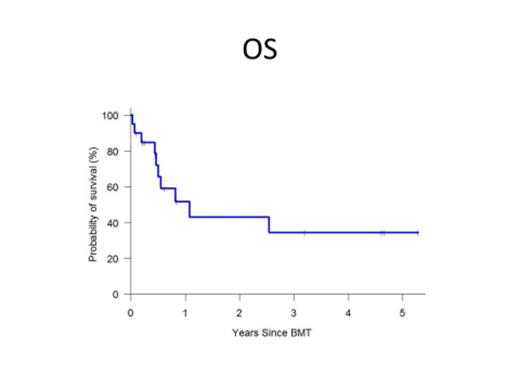
All patients achieved sustained engraftment with median times to neutrophil and platelet engraftment of 17.5 (14-44) days and 32 (15-99) days respectively. The cumulative incidences (CI) of grade II-IV and grade III-IV acute GVHD at 180 days were 36% and 10% respectively. The CI of moderate-severe chronic GVHD was 13% at 1 year post HIDT. At a median follow-up of X months, The probability of overall survival, DFS, NRM and relapse post HIDT were 52%, 39%, 29% and 33% at 1 year and 34%, 31%, 29% and 40% at 3 years respectively.
Conclusions:
HIDT is an effective strategy to treat relapsed hematologic malignancies after a matched sibling or matched unrelated donor HSCT. Further and larger cohorts to confirm these observations are warranted.
Survival Estimates
| . | 6 months . | 1 year . | 3 years . |
|---|---|---|---|
| OS | 72% | 52% | 34% |
| DFS | 53% | 39% | 31% |
| NRM | 22% | 29% | 29% |
| Relapse | 25% | 33% | 40% |
| Mod-severe chronic GVHD | 5% | 13% | 13% |
| Grade II-IV acute GVHD | 36% | ||
| Grade III-IV acute GVHD | 10% |
| . | 6 months . | 1 year . | 3 years . |
|---|---|---|---|
| OS | 72% | 52% | 34% |
| DFS | 53% | 39% | 31% |
| NRM | 22% | 29% | 29% |
| Relapse | 25% | 33% | 40% |
| Mod-severe chronic GVHD | 5% | 13% | 13% |
| Grade II-IV acute GVHD | 36% | ||
| Grade III-IV acute GVHD | 10% |
1. Christopeit M, Kuss O, Finke J, Bacher U, Beelen DW, Bornhauser M, et al. Second allograft for hematologic relapse of acute leukemia after first allogeneic stem-cell transplantation from related and unrelated donors: the role of donor change. J Clin Oncol. 2013;31(26):3259-71.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal