Abstract

Introduction
Autologous transplantation is considered standard therapy for young and fit myeloma patients after initial therapy. Cyclophosphamide+ G-CSF is considered standard therapy for collection even though there is evidence for minimal anti-myeloma effect of cyclophosphamide, some increased short term toxicity and potential concerns regarding long term toxicity. There have been a few retrospective comparisons and one randomized study comparing cyclophosphamide based and G-CSF alone based PBSC collection. To our best knowledge they have not reported any impact in progression free survival (PFS) or overall survival (OS). We have compared here our myeloma patient cohort to explore these important endpoints.
Patients and methods
89 patients (55 male and 34 female) who underwent first autologous transplant for myeloma between 2003 and 2010 were analysed in our study. Mobilization was with G-CSF alone in 45 patients (median age 58 yrs, range 38-70 yrs.) and cyclophosphamide and G-CSF in 44 (median age 58 yrs, range 41-74 yrs.). Cyclophosphamide was used at 3g/m2 and in both cases G-CSF used was lenogastrim at 10mcg/kg. There were 7 patients with ISS stage 3 in the G-CSF only group as compared to 10 in the Cyclophosphamide group. Prior chemotherapy was cyclophosphamide, thalidomide and dexamethasone in majority of cases (n=55) with no difference across both groups. Data regarding high risk genetics and pre-transplant response was unavailable. We collected data progression free survival, overall survival, harvest dose and engraftment kinetics. Data was analyzed using SPSS and log rank test.
Results
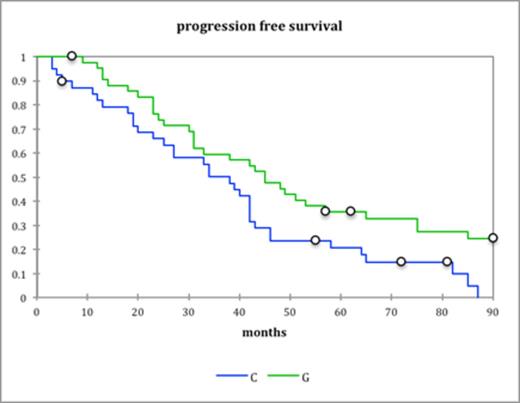
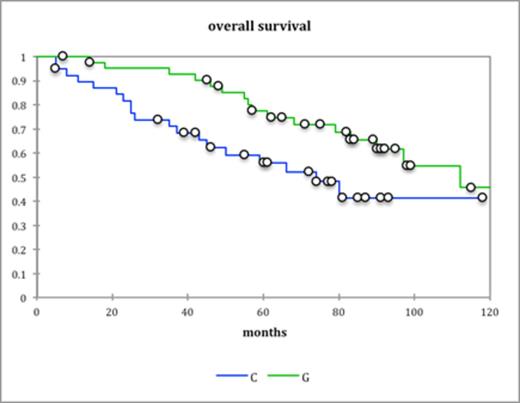
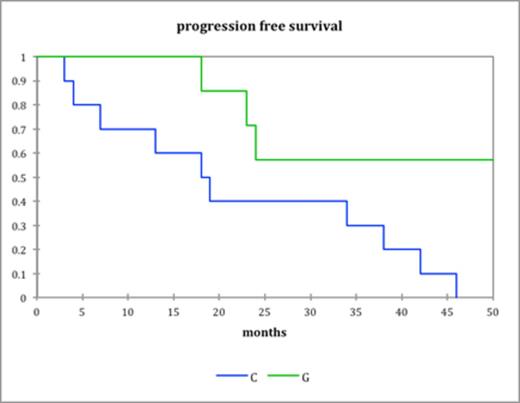
The median dose of stem cells collected with G-CSF alone was 3.59×106 CD34 cells/kg (range 1.84-8.09) where as with cyclophosphamide and G-CSF it was 3.8×106 CD34 cells/kg (range 1.6-13). There was no difference in engraftment between the two groups with median neutrophil engraftment (Absolute neutrophil count>0.20×109/L) at day 14 and platelet engraftment (>50×109/L) at day 15 and 16 respectively. Progression free survival was significantly better in G-CSF alone cohort (46 months vs. 38 months, P=0.016) (fig. a) Overall survival was better in the GCSF only group as well (113 months vs. 75 months, P=0.029) (fig b). In the 17 high risk patients PFS was much better in the G-CSF group (60months vs. 22 months, P=0.02) (Fig c). There were 4 (9%) admissions in the cyclophosphamide group due to neutropenic sepsis as compared to none in the G-CSF group.
Discussion
Cyclophosphamide and G-CSF may be associated with slightly higher stem cell yields but this margin is becoming smaller and not significant in the era of liberal plerixafor usage. In addition some patients are hospitalized due to neutropenic sepsis with this regimen. Our data shows anti-myeloma effects of cyclophosphamide +G-CSF is not demonstrated. There are ongoing studies from the Finland group which show no difference in the number of CD34+ cells collected after initial therapy with lenalidomide. The only difference is the number of days required for apheresis. In addition to above our single center experience shows both PFS and OS benefit for G-CSF only PBSC mobilization. . This may partially be explained by the slight difference in ISS risk stages in our patients but on censoring for ISS stage 3 these results were more pronounced. This is the first time we have seen any report point out towards a PFS and OS difference between two widely used mobilization regimens. This needs testing in a large randomized multi-center study to see if there is a difference and if so is this due to a change in milieu of lymphocytes. We have previously reported that absolute lymphocyte count on day 15 post autograft was reflective of a higher lymphocyte count in the apheresis bag in case of G-CSF only mobilizations as compared to cyclophosphamide +G-CSF. The absolute lymphocyte count on day 30 was a predictor for better OS.
Sutton:bayer: Honoraria. Paneesha:Janssen: Consultancy. Nikolousis:Alexion: Honoraria. Kishore:Jazz pharma: Honoraria; Celgene: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal