Abstract
Background: Burkitt Lymphoma (BL) is the most common form of NHL in children and adolescents and has an excellent prognosis (≥80% 5years, EFS, Cairo et al. Blood, 2007, Cairo et al. JCO, 2012). The prognosis has improved with the addition of targeted immunotherapy with rituximab (Goldman/Cairo et al, Leukemia, 2013, Goldman/Cairo et al. BJH, 2014). However, a subset of patients with chemoimmunotherapy-resistant disease has a dismal prognosis (≤ 10% 5 years, EFS) (Miles/Cairo et al. BJH, 2012). Deregulation of signaling pathways controlled by protein phosphorylation underlies the pathogenesis of B-cell lymphomas, however, the extent to which they contribute to rituximab resistance is largely unknown (Barth et al. BJH, 2013). Obinutuzumab (GA101), a novel glycoengineered type II CD20 Ab vs. RTX, a Type I CD20 Ab, mediates enhanced cell death & ADCC against diffuse B-cell lymphoma vs. RTX (Mössner et al. Blood, 2010), and was recently approved by FDA and EMA for first line treatment of CLL in combination with chlorambucil.
Objective: To evaluate phosphorylation of signaling pathways are differentially altered following obinutuzumab vs RTX against RTX-sensitive/resistant BL.
Methods: Raji (CD20+, ATCC, Manhass, VA) and Raji-4RH (provided by M. Barth, Roswell Park Cancer Institute) were cultured in RPMI with 10% FBS. For in-vitro studies, tumor cells were incubated with 100 µg/ml obinutuzumab (supplied by Christian Klein, PhD, Roche Research & Early Development, Zurich), and/or RTX for 24 hrs. For Phosphoproteomics analysis, we performed a mass spectrometry-based label-free quantitative phosphoproteomic profiling of the BL cell lines Raji/Raji4RH in the presence/absence of obinutuzumab or rituximab (100µg/ml for 24h) or isotype control. Six milligrams of protein from each condition were digested by trypsin and peptides and subjected to phosphopeptide enrichment using metal oxide affinity chromatography (MOAC) and immunoprecipitation. An LTQ Orbitrap XL, in-line with a Paradigm MS2 HPLC was employed for acquiring high-resolution MS and MS/MS data that were searched with the Swissprot Human taxonomic protein database (McDonnell and Lim et al, Blood, 2013). Silencing of PLCG2 in Raji and Raji4RH cell lines was carried out according to the manufacturer's instructions (Dharmacon, PA, USA).
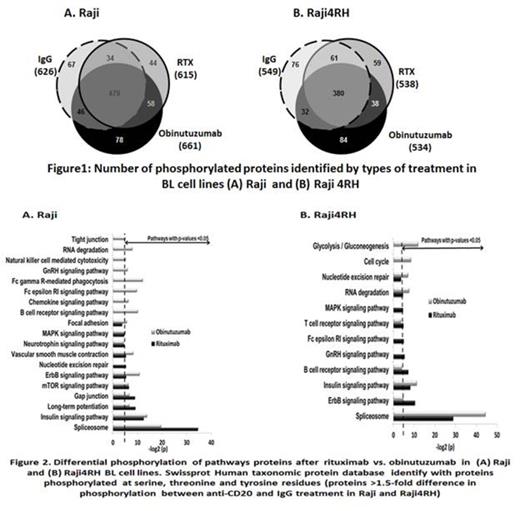
Results: Four hundred and eighteen out of total 661 proteins in Raji and 377 out of total 534 proteins in Raji4RH were differentially phosphorylated (>1.5 fold) after obinutuzumab treatment. Of these proteins, 46 were expressed at significantly higher levels in obinutuzumab vs. RTX in Raji (Figure 1). However, Raji4RH, the RTX resistant cell line did not show significant increase in phosphorylation of protein following in obinutuzumab vs. RTX. Proteins differentially phosphorylated in response to obinutuzumab vs. RTX are involved in the BCR (PLCG2, BTK, GSK3B and RAF-1), FC gamma phagocytosis (FCRG2B, MAPK1, PLCG2 and RAF-1), and Natural Killer cell-mediated cytotoxicity (MAPK1, RAF-1, PLCG2 and MAPK3) signaling pathways in Raji vs. Raji4RH (Figure 2). Differential phosphorylation of BCR signaling pathways proteins (BTK, PLCG2 and GSK3B), validated by western blot studies after incubation with obinutuzumab vs. RTX in Raji/Raji4RH cell lines, revealed up-regulation of BTK and PLCY2 after obinutuzumab treatment vs. RTX treatment in Raji BL cell line. Silencing one of the BCR signaling pathway protein, PLCG2 significantly increased cell proliferation and decreased cell death after obinutuzumab vs. RTX treatment in Raji (P=0.0001 & 0.004) however, there was no change in Raji 4RH RTX resistant cell line.
Conclusions: Obinutuzumab and RTX differentially phosphorylate BCR, phagocytosis and cytotoxicity signaling pathways in BL. Knockdown of PLCG2 significantly enhanced BL proliferation and reduced cell death after obinutuzumab vs. RTX treatment. These results offer insights into alternate therapeutic strategies in the treatment of RTX resistant BL.
Klein:Roche: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal