Abstract
Background: Peripheral T-Cell lymphomas (PTCLs) constitute approximately 10% of lymphoid malignancies and consist of several distinct entities based on pathologic and clinical characteristics. With the exception of a few subtypes (e.g., ALK-positive anaplastic large cell lymphoma (ALCL) and some primary cutaneous or leukemic forms of PTCL), a majority of PTCLs are aggressive as characterized by poor treatment response, rapid disease progression and poor overall survival. We have shown that landmark timepoints of event-free survival after diagnosis can stratify subsequent overall survival (OS) in diffuse large B-cell and follicular lymphoma. Here we evaluate this approach in newly diagnosed aggressive PTCLs treated with anthracyline-based or related chemotherapy.
Methods. Newly diagnosed PTCL patients were prospectively enrolled in the University of Iowa/Mayo Clinic Lymphoma SPORE Molecular Epidemiology Resource (MER) from 2002-2012. Clinical data were abstracted from medical records using a standard protocol. For this analysis, we included patients receiving anthracycline-based or other multiagent chemotherapy for the following PTCL subtypes: ALK-negative ALCL (N=24); angioimmunoblastic T-cell lymphoma (AITL, N=34); PTCL, not otherwise specified (NOS; N=60); enteropathy-associated T-cell lymphoma (EATL, N=8); extranodal NK/T-cell lymphoma, nasal type (ENKTL, N=11); and hepatosplenic T-cell lymphoma (HSTCL, N=1). Patients were prospectively followed, and event-free survival (EFS) was defined as time from diagnosis to progression, re-treatment, or death due to any cause. Landmark EFS timepoints were assessed at 12 (EFS12) and 24 (EFS24) months after the date of diagnosis. Subsequent OS was defined as time from a specific endpoint (diagnosis, event or EFS landmark). Replication was performed in a population-based cohort of T-cell lymphomas diagnosed from 2000-2009 from the Swedish Lymphoma Registry.
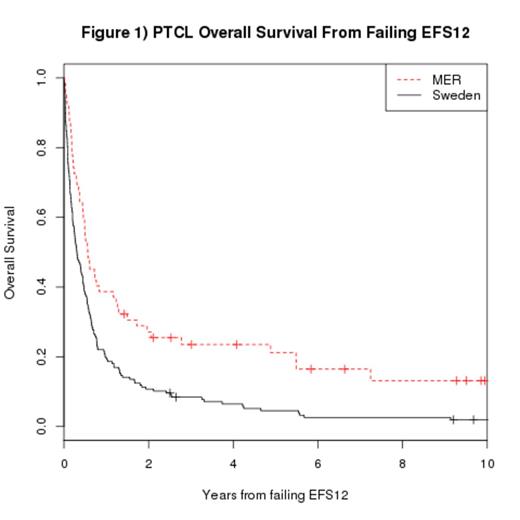
Results. 138 eligible patients were enrolled in the MER from 2002-2012, the median age at diagnosis was 58 years (range, 19-88), 66% were male, 73% had Stage III-IV disease, and 33% had IPI 0-1. At a median follow-up of 47 months (range 11-120), 87 patients (63%) had an event and 70 patients (51%) had died. From diagnosis, only 60 patients were event-free at 12 months (EFS12 45%). Patients who failed to achieve EFS12 had a poor subsequent OS from event (median OS = 6.8 months, 95% CI: 5.3-14.0, figure 1). In contrast, patients who achieved EFS12 had a favorable subsequent OS (median unreached, figure 2). Of the 427 eligible patients in the Swedish registry, the median age at diagnosis was 66 years (range, 18-88), 63% were male, 68% had Stage III-IV disease, and 25% had IPI 0-1. PTCL subtypes were: ALK-negative ALCL (N=89); AITL (N=80); PTCL, NOS (N=183); EATL (N=44); ENKTL (N=24); and HSTCL (N=7). At a median follow-up of 86 months (range 40-158), 333 patients (79%) had an event and 316 patients (74%) had died. From diagnosis, 183 patients were event-free at 12 months (EFS12 44%). Similar to the MER cohort, Swedish patients failing EFS12 had poor subsequent survival (median OS = 3.7 months, 95% CI: 2.9-5.3, figure 1). Swedish patients achieving EFS12 had a favorable subsequent OS (median OS = 89 months, figure 2). Similar results were obtained when conducting landmark analysis at 24 months after diagnosis (EFS24).
Conclusion. Relapse and re-treatment events within the first 12 months of diagnosis are associated with very poor OS in PTCL treated with anthracyclines or related chemotherapy, while patients achieving EFS12 have encouraging subsequent OS. Stratifying patients into prognostically distinct subsets using EFS12 may help focus biologic and biomarker studies. EFS12 has potential as an early endpoint for studies of newly diagnosed PTCL. Further investigation of determinants related to post-EFS12 survival is needed.
Maurer:Kite Pharma: Research Funding. Cerhan:Kite Pharma: Research Funding. Ansell:Bristol-Myers Squibb: Research Funding; Celldex: Research Funding. Link:Genentech: Consultancy, Research Funding; Kite Pharma: Research Funding. Thompson:Kite Pharma: Research Funding. Relander:Respiratorius: Patents & Royalties: valproate for DLBCL.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal