Abstract
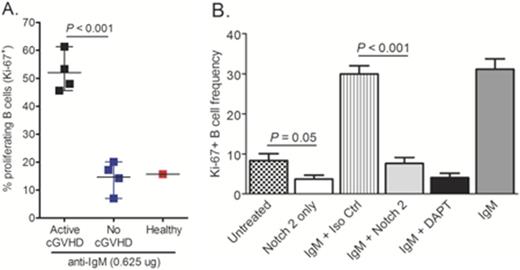
While Notch signaling is being well studied with regard to T cell pathology and graft-versus host disease (GVHD) (Tran IT et al., 2013. J. Clin. Invest.), the role of Notch receptors in the development and activation of B cell subsets in chronic GVHD (cGVHD) genesis remains unknown. We previously identified a subset of Ôpre-germinal centerÕ B cells within the peripheral blood of cGVHD patients that is largely absent in patients without cGVHD. In addition to cell surface characteristics, this extrafollicular B cell subset has potential functional characteristics of marginal zone (MZ)-like B cells, including increased responsiveness to surrogate antigen stimulation. Along with increased proliferative responses to BCR stimulation, B cells from patients with active cGVHD had significantly increased signaling via proximal B cell receptor (BCR) molecules, including Syk and BLNK. In murine models with lymphopenic environments, Notch 2 binds the ligand Delta-like 1 (DLL1/Dll1) and drives maturation of MZ-like B cells. Also, healthy human B cells have increased Notch receptor responsiveness after BCR stimulation. Together previous studies allowed us to hypothesize that a Notch 2 signaling axis underpins B cell hyper-responsiveness in human cGVHD. We found that limiting dose BCR stimulation with surrogate antigen in the presence of Notch ligand over-expressing cells (OP9-DL1) resulted in maintenance of cell surface Notch 2 expression at significantly higher levels on B cells from patients with active cGVHD compared to patients without cGVHD, as assessed by flow cytometry analysis (P < 0.01). We also found that in the presence of Notch ligand, B cells from patients with active cGVHD responded to minimal BCR stimulation with surrogate antigen. Using nearly 100x less surrogate antigen than was required to induce proliferation without Notch ligand, cGVHD B cells proliferated to a significantly greater degree than B cells from patients with no cGVHD, as evaluated by Ki-67 staining using flow cytometry (P < 0.001 in a two-sided t-test, Figure 1A). Likewise, concomitant BCR- Notch activation of active cGVHD patient B cells was associated with significantly increased B-cell size compared to patients without disease (P < 0.01). BLNK expression in active cGVHD B cells was also maintained at higher levels under these conditions, suggesting a mechanistic link between the BCR and Notch pathways in cGVHD. Strikingly, targeting Notch 2 with an antagonistic monoclonal antibody (mAb) (Wu Y et al., 2010. Nature; kindly provided by Genentech, Inc.) completely abrogated the BCR-Notch axis hyper-responsiveness of active cGVHD B cells without affecting B-cell survival (P < 0.001, Figure 1B). In this in vitro system, using nanoString Technologies¨ gene profiling, we found that two, well-defined effector genes downstream of Notch signaling were significantly decreased in active cGVHD B cells after exposure to the anti-Notch 2 mAb (P = 0.0006 and P < 0.02, respectively, compared to isotype control mAb). Also consistent with a Notch 2-driven activation pathway, the expression of multiple genes involved in homeostasis/cell cycle regulation were altered in active cGVHD B cells exposed to anti-Notch 2 mAb (P < 0.01). Finally, ongoing in vivo analyses of the Notch 2 mAb in a pre-clinical mouse model of cGVHD indicates that Notch 2 blockade does not negatively impact early B cell recovery following bone marrow transplantation. These results may reveal that therapeutic targeting of Notch 2 alone would be sufficient to quell B cell hyper-responsiveness in active cGVHD, while preserving protective humoral immunity.
In summary, our data suggest a working model in which Notch-mediated aberrant B cell maturation contributes to cGVHD pathophysiology. In this model, Notch 2 stimulation along with a combination of complex B-cell selection and tolerance mechanisms afford production of pathological B cells. Given that Notch 2 is a cell surface receptor expressed by activated B cell subsets of pathological relevance, and Notch 2 blockade has been shown to be well-tolerated in pre-clinical models, our findings support an important clinical opportunity: Targeting Notch 2 on B cells in active cGVHD represents a viable future therapeutic strategy worthy of continued investigation.
This work was supported by National Institutes of Health grant 5K08-HL107756, and a Translational Research Program grant from the Leukemia & Lymphoma Society.
Rizzieri:Teva: Other: ad board, Speakers Bureau; Celgene: Other: ad board, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal