Abstract
Background: The detection of minimal residual disease (MRD) following therapy in acute myeloid leukemia (AML) is associated with increased risk of relapse and poor survival and can influence clinical decisions regarding treatment. Multi-parameter flow cytometry can assess MRD in ~90% of AML cases (due to aberrant surface antigen expression); however, its poor sensitivity typically requires bone marrow aspirates, which are invasive and cannot be obtained frequently. Polymerase chain reaction (PCR) assays are highly sensitive in detecting AML-associated mutations, but significant sample processing and assay development are needed to detect patient-specific mutations, which curbs the broad applicability of PCR for the majority of AML patients. Here we present a microfluidic chip assay (Figure 1) with high sensitivity for detecting low levels of aberrant antigen expressing blasts in peripheral blood, thereby enabling frequent MRD monitoring for high-risk AML patients undergoing allogeneic stem cell transplant (SCT). Early detection of MRD in AML post-SCT can influence clinical decisions, such as adjustments in immunosuppression or administration of donor lymphocytes.
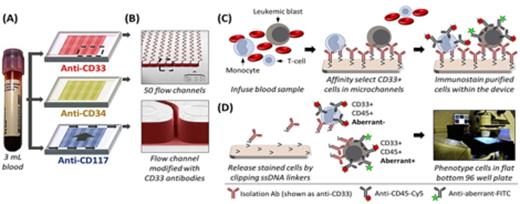
Methods: Subjects had 3 mL of peripheral blood drawn into an EDTA tube every 2 weeks following their SCT from day 14 through day 98 and monthly through day 365 post-SCT. The blood was injected into microfluidic chips (Figure 1), each containing 50 flow channelsthat were chemically modified with capture antibodies specific for human CD34, CD33 or CD117 - surface antigens commonly expressed by AML blasts. Following cell selection, fluorescently-labeled antibodies specific for 1) the capture antibody, 2) an aberrant lineage marker (e.g. CD7, CD56) that was known to be present on the patients AML blasts and 3) CD45, were injected into the chips. Cells were then released for off-chip fluorescence microscopy and enumerated with the results compared to conventional MRD assays, ongoing treatments and clinical outcomes.
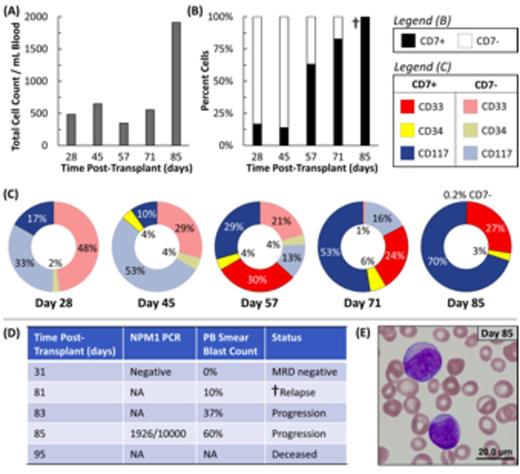
Results: 7 patients were enrolled with 4 having reportable data at the time of submission. Pt#1 had AML with NPM1 and FLT3-ITD mutations. NPM1 PCR classified Pt#1 as MRD(-) 51 days after SCT, while the microfluidic assay was MRD(+) at day 48 with 81 aberrant cells/mL. The aberrant cell count began to steadily increase at day 77 that persisted until Pt#1 was diagnosed with hematologic relapse on day 85 and died at day 95 (Figure 2). Pt#2 is currently 216 days post-SCT and has had persistent low levels (5 - 57 cells/mL) of aberrant cells since day 64. This patient recently developed GVHD and is being treated with corticosteroids and has no evidence of hematologic relapse at this time. Pt#3 developed high level aberrant cells (1066 cells/mL) at day 35 post SCT. These values dropped while the patient was treated with valganciclovirfor CMV viremia (22 cells/mL at day 47), but the high level of AML blasts returned upon completion of therapy (443 cells/mL at day 105). This patient developed multi-organ system failure and at the time of death (day 124), circulating immature cells were detectable in the peripheral blood. Pt#4 has AML with an NPM1 type A mutation. The patient was MRD(+) as determined by NPM1 PCR analysis (checked 3 times) and by the microfluidic assay (checked 7 times); however, on the most recent assessment at day 125, both assays were MRD(-).
Conclusion: A microfluidic assay was able to isolate trace quantities of circulating AML blasts (<10 cells/mL) and could be used to frequently assess MRD, showing high concordance with therapeutic treatment and conventional diagnostics. For one patient, the microfluidic assay detected MRD earlier than the NPM1 PCR analysis. The assay used only 3 mL of peripheral blood with no special blood handling or pre-processing required, making it suitable for frequent disease status monitoring. Further research will involve integrating the microfluidic chips with a microfluidic flow cytometer to further improve the speed and efficiency of detecting circulating AML blasts.
(A-B) Microfluidic chips and (C-D) assay workflow for frequent MRD analysis in AML. A single blood sample was split 3-ways so that each chip processed the same volume.
(A-B) Microfluidic chips and (C-D) assay workflow for frequent MRD analysis in AML. A single blood sample was split 3-ways so that each chip processed the same volume.
Disease progression for Pt#1 determined by (A-C) the microfluidic assay and (D-E) conventional diagnostics.
Disease progression for Pt#1 determined by (A-C) the microfluidic assay and (D-E) conventional diagnostics.
Soper:Biofluidica: Membership on an entity's Board of Directors or advisory committees, Patents & Royalties.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal