Abstract
BACKGROUND. A set of Quality Controls (QC) of a Cord Blood Unit (CBU) at release is required by International Standards for Cord Blood banking, often including Flow Cytometry (FC) counting and viability assessment of thawed stem cells. Reliability and reproducibility of such tests across different CB banks is still an unmet need. Indeed, discrepancies between the thawed product characterized at the Transplant Center and the QC performed at the CB bank, might be addressed to different assessment technologies. Therefore, there is room for a multicenter exercise, aimed to check the reproducibility of the flow cytometry-driven QC and possibly to propose technical standardization and improvements.
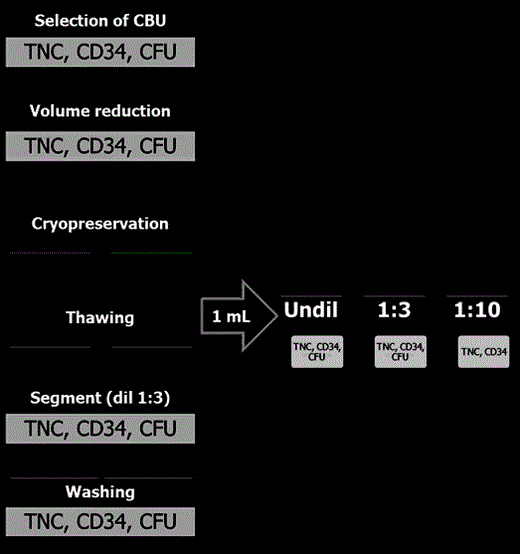
In this context, a protocol for manipulation and FC assessment of thawed CB samples was circulated and shared among 5 European CB banks(Dusseldorf, Barcelona, Florence, Cremona, Nottingham) , on behalf of Eurocord and Netcord.
METHODS. Different techniques of sample preparation were tested on thawed samples, washed samples and the segment attached to the CB bag, respectively, as shown in Fig. 1. A solution of Dextran and Human Albumin as described by the New York CBB was used for dilution/washing the samples. In particular, one sample of each thawed Unit was processed for FC assay undiluted and diluted 1:3 and 1:10, respectively. Five CB Units (CBU) per CB bank unsuitable for banking (with TNC count≥1x109) were selected for the assay.
The FC assay protocol was a standard TruCOUNT®, ISHAGE-based (SI), single-platform multigating strategy for CD34+ cells counting, shared by all the Banks in order to standardize the gating strategy. A mock sample was circulated before the exercise in order to check the assay reproducibility. A modification to the ISHAGE methodology (MI) was also proposed with the aim of improving the assessment of CD34+ cells viability in thawed samples.
RESULTS A total of 25 CBU across the 5 labs were cryopreserved and thawed following local standards; pre-freezing and post-thawing samples were assessed for CD34+ recovery and viability.
Data from all Banks were centralized and statistical analysis was performed.
TNC recoveries resulted to be reproducible in all Centers for all the thawed products (undiluted, 1:3 and 1:10 dilution, segment 1:3 diluted and washed undiluted samples, respectively).
Both SI and MI strategies showed no significant discrepancies when determining CD34+ absolute number and viability in pre-freezing CBUs (p>0.05 with pair T test).
The recovery of viable CD34+ cells assessed by SI or MI in the thawed products did not show any significant difference in all the experimental conditions, whilst the recovery of total CD34+ cells was always lower in SI-analyzed samples than in MI-analyzed samples (* = pair T test p<0,05). One to ten dilution of the thawed product showed a higher variability, as well as segment data when compared to the bag, likely for the small size of the sample.
Post-thawing CD34+ viability in MI methodology was significantly lower than in SI technique in all the experimental conditions (Fig.2).
Finally, CFU recovery did not show significant differences in post thaw 1:3, washed 1:1 and segment 1:3 diluted samples when related to the pre-freeze clonogenic potential. Interestingly, MI showed a better correlation with CFU.
CONCLUSIONS
· Flow Cytometry-based assessment of CB graft before and after cryopreservation can provide reproducible results, following an adequate standardization of sample preparation and gating strategy.
· The segment-related data were shown to be a reliable Quality Control of the stored CB Unit
· Standard ISHAGE technology was less reliable in the CD34+ cell viability assessment in the thawed samples and might therefore lead to an overestimation of CD34+ viability.
An adaptation of both the acquisition setting and the gating strategy is needed in order to detect viable and non viable CD34+ cells in thawed CB units. The dissemination of these findings might have a significant impact in CB transplantation, as a validated and standardized, single platform, CD34+ cell count methodology is an essential step to reduce the intra- and inter-laboratory variability. In particular, such standardization might reduce the discrepancies between QC at release in the CB banks and the thawed product at bedside.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal