Abstract
Background: Diagnostic and therapeutic advances have significantly improved the outcomes for multiple myeloma (MM) patients. However, pts who are refractory to or relapse after therapy with immune modulatory drugs and proteasome inhibitors remain a therapeutic challenge. Comprehensive genomic profiling via clinical next generation sequencing (NGS)-based assays studies of MM cases have revealed multiple targetable mutations that were previously unexploited in MM.
Methods: Between June 2013 and May 2014 we performed genomic profiling of 351 patients who had progressed after initial therapy to assist physicians in therapy planning. Comprehensive genomic profiling was performed using the FoundationOne¨ or FoundationOne Heme¨ assays. FoundationOne assays 374 cancer-related and 24 frequently rearranged genes via DNA-seq, and FoundationOneHeme assays 405 cancer-related and 31 frequently rearranged genes via DNA-seq as well as 265 frequently rearranged genes by RNA-seq. All samples were sequenced in a CLIA-certified CAP-accredited laboratory to an average depth >500x .
Patients with activating alterations of KRAS, NRAS or BRAF were considered for therapy with the targeted agent trametinib (TMTB) as were patients who had a gene expression signature suggesting activation of the MAPK pathway. Retrospective review of this case series was approved by the UAMS institutional review board.
Results: We identified 63 patients who underwent treatment with Trametinib. 60 were treated based on activating mutations of KRAS, NRAS or BRAF and 3 were treated based on a GEP signature. The median age was 65 and patients had a median of 5 lines of prior therapy (range 1-20). 38 of 63 patients had prior treatment with Total Therapy. 43 underwent salvage with chemotherapy prior to initiation of TMTB, 15 had salvage transplants, 33 patients were exposed to novel agents (Pomalidomide, Carfilzomib) and 33 had Metronomic therapy before TMTB. 25% of patients were ISS stage 3 and 37% had GEP70 defined high risk. 13 had PET defined extra medullary disease (EMD).
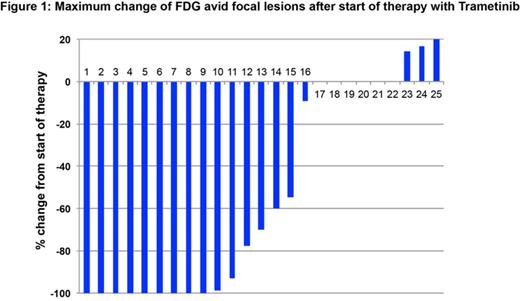
41 patients were administered TMTB monotherapy and 22 received TMTB treatment in combination with other agents. In general the treatment was well tolerated. 10 patients discontinued therapy because of toxicities, 29 discontinued because of disease progression or death. None of the deaths were attributed to TMTB, Best treatment responses were SD in 30, PR in 8, VGPR in 2 and CR in 3 of the 63 pts. For 25 patients with evaluable PET data, treatment resulted in complete resolution of FDG avid lesions in 9 patients and a better than 50% reduction in 15 (Figure 1). We will present updated data on clinical responses as well as toxicities.
Conclusions: Treatment with targeted therapy guided by prospective comprehensive genomic profiling across all classes of genomic alterations in this heavily pretreated population of MM patients resulted in an unexpectedly high objective response rate. Observation of CR with TMTB monotherapy further supports continued investigation of this individualized approach to MM management.
Van Laar:Signal Genetics: Employment, Equity Ownership. Ali:Foundation Medicine, Inc.: Employment, Equity Ownership. Miller:Foundation Medicine, Inc: Employment. Zangari:Norvartis: Membership on an entity's Board of Directors or advisory committees; Onyx: Research Funding; Millennium: Research Funding. van Rhee:Millenium: Speakers Bureau; Sanofi: Speakers Bureau; Celgene: Speakers Bureau; Janssen: Speakers Bureau. Morgan:Celgene Corp: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Myeloma UK: Membership on an entity's Board of Directors or advisory committees; International Myeloma Foundation: Membership on an entity's Board of Directors or advisory committees; The Binding Site: Membership on an entity's Board of Directors or advisory committees; MMRF: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal