Abstract
OFA was approved for the treatment of fludarabine and alemtuzumab refractory CLL on the basis of a single-arm study which also included pts with BFR CLL. This open label, randomized, Phase III study of OFA or PC treatment in pts with CLL refractory to fludarabine and with bulky lymphadenopathy (at least one lymph node > 5 cm) was conducted to confirm the results of OFA in BFR pts and also through a 2nd randomization to compare extended OFA (12 months) vs. approved regimen of OFA (6 months). PC was considered an appropriate comparator for these pts given the lack of a consensus around standard of care treatment for this difficult-to-treat population.
Pts were randomized 2:1 to receive either the approved regimen of 8 weekly infusions of OFA followed by 4 monthly infusions (Dose 1, 300 mg; Doses 2–12, 2000 mg) or PC of non-OFA containing therapy, including treatments approved for CLL and well established standards of care for up to 6 months. Pts in the OFA arm who had an investigator-assessed CR, PR or SD underwent a 2nd 2:1 randomization to 6 additional OFA infusions 2000 mg every 4 weeks (OFA Ext), or observation (Obs). Pts in the PC arm who developed disease progression during the study could receive OFA salvage therapy for up to 12 months of treatment. Premedication for OFA infusions consisted of glucocorticoid, paracetamol, and antihistamine.
The primary objective of this study was to evaluate progression-free survival (PFS) with OFA treatment versus PC as assessed by an Independent Review Committee (IRC). Response assessments were performed using the 2008 Update of the NCI-WG CLL Guidelines [Hallek, 2008]. Secondary endpoints included overall response rate (ORR), time to next therapy (TNT), overall survival (OS), and safety.
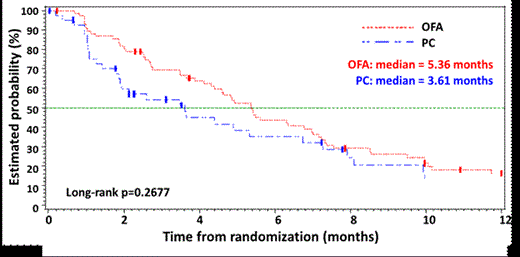
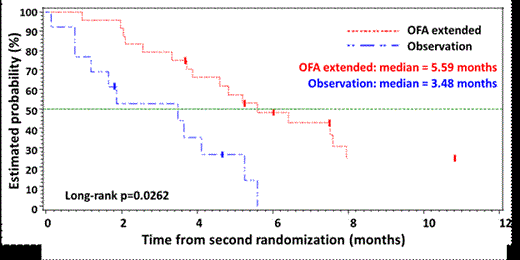
122 pts were randomized (79 OFA, 43 PC). Pts completed a median of 12 OFA infusions and 3 months PC therapy. The median PFS (mPFS) as measured by IRC was 5.4 months for OFA and 3.6 months for PC (hazard ratio [HR] 0.79, p=0.27, stratified log rank test; Fig 1). Median PFS as assessed by the investigator (INV) was 7.0 months for OFA and 4.5 months for PC (HR 0.56, p=0.003). The median time to start of next anti-CLL treatment was 11.5 months for OFA and 6.5 months for PC (p=0.0004, stratified log rank test). The ORR (95% CI) by IRC evaluation was 38% (27%, 50%) for OFA and 16% (7%, 31%) for PC (p=0.02). 37 pts underwent a 2nd randomization to either OFA Ext (n=24) or Obs (n=13). From 2nd randomization, mPFS (INV) was 5.6 months for OFA Ext and 3.5 months for Obs (HR: 0.49, p=0.026, stratified log rank test) (Fig 2). Grade ³3 infusion reactions occurred in 5% of pts in the OFA arm with no fatal reactions in either arm. Grade ≥3 cytopenias in the OFA arm included neutropenia (24%, no prolonged neutropenia and 1 patient with late onset neutropenia), thrombocytopenia (8%), anemia (8%). Of the 43 PC pts, 22 received OFA Salvage with an ORR of 50% and mPFS of 5.4 months.
Table of Response and Time to event Endpoints
| . | OFA (N=79) . | PC (N=43) . | ||||
|---|---|---|---|---|---|---|
| Characteristics | Median (range) | |||||
| Age, years | 61.5 (46 – 82) | 63 (40 – 76) | ||||
| No. prior therapies | 4 (2 – 16) | 3 (2 – 11) | ||||
| No. of infusions | 12 (1 – 18) | 3 (1 – 6) | ||||
| IRC | Investigator | |||||
| Efficacy endpoints | OFA | PC | p-value | OFA | PC | p-value |
| Responders, % | 38% | 16% | 0.019 | 49% | 37% | 0.415 |
| mPFS, months | 5.36 | 3.61 | 0.267 | 7 | 4.5 | 0.003 |
| OFA | PC | |||||
| TNT, months | 11.5 | 6.5 | ||||
| p=0.0004 | ||||||
| OS, months | 19.2 | 14.5 | ||||
| p=0.13 | ||||||
| . | OFA (N=79) . | PC (N=43) . | ||||
|---|---|---|---|---|---|---|
| Characteristics | Median (range) | |||||
| Age, years | 61.5 (46 – 82) | 63 (40 – 76) | ||||
| No. prior therapies | 4 (2 – 16) | 3 (2 – 11) | ||||
| No. of infusions | 12 (1 – 18) | 3 (1 – 6) | ||||
| IRC | Investigator | |||||
| Efficacy endpoints | OFA | PC | p-value | OFA | PC | p-value |
| Responders, % | 38% | 16% | 0.019 | 49% | 37% | 0.415 |
| mPFS, months | 5.36 | 3.61 | 0.267 | 7 | 4.5 | 0.003 |
| OFA | PC | |||||
| TNT, months | 11.5 | 6.5 | ||||
| p=0.0004 | ||||||
| OS, months | 19.2 | 14.5 | ||||
| p=0.13 | ||||||
Kaplan-Meier Estimates of PFS by IRC (OFA vs. PC)
Kaplan-Meier Estimates of PFS by INV (OFA Ext vs. Obs from 2nd randomization)
Kaplan-Meier Estimates of PFS by INV (OFA Ext vs. Obs from 2nd randomization)
Conclusions: Although the study did not meet its primary endpoint of demonstrating statistically significant superior PFS by IRC, the ORR, PFS by investigator, TNT, and OS favor OFA and were consistent with previously reported results in BFR pts. There was also a longer PFS in pts who underwent the 2nd randomization to receive OFA Ext vs. the approved OFA treatment regimen of 6 months treatment.
Österborg:GSK: Research Funding. Off Label Use: Discussion of extended ofatumumab dosing (12 months) in addition to the approved 6 month dosing of ofatumumab in refractory CLL. Zaritskey:Novartis: Consultancy, Honoraria. Kaplan:GSK: Honoraria. Steurer:GSK: Consultancy, Honoraria, Research Funding. Schuh:GSK: Honoraria; Roche: Honoraria; Gilead: Honoraria; Celgene: Honoraria; Napp: Honoraria. Montillo:GSK: Honoraria. Kulyaba:GSK: Honoraria. Gorczyca:GSK: Employment. Daly:GSK: Employment. Chai-Ni:GSK: Employment. Lisby:Genmab: Employment. Gupta:GSK: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal