Abstract
Post transplant thrombotic microangiopathy (PT-TMA) has been recognized for over 25 years. The exact incidence of PT-TMA in small bowel transplant (SBT) patients is unknown with reports varying from 4% to greater than 30%. One of the contributors to the differences in reported occurrence rates is the continued debate regarding the definition of PT-TMA. There is a growing consensus that PT-TMA is complement mediated and therefore can be defined by the same criteria as atypical hemolytic uremic syndrome (aHUS). AHUS is a complement mediated TMA defined by microangiopathic hemolytic anemia, thrombocytopenia (TCP), and end organ damage. We have previously demonstrated that the initiation of eculizumab in post-transplant aHUS (PT-aHUS) has led to improved outcomes in these patients. We believe that establishing reliable diagnostic criteria that can be utilized in the post transplant period will lead to earlier recognition and treatment which will decrease the high mortality rate associated with PT- aHUS.
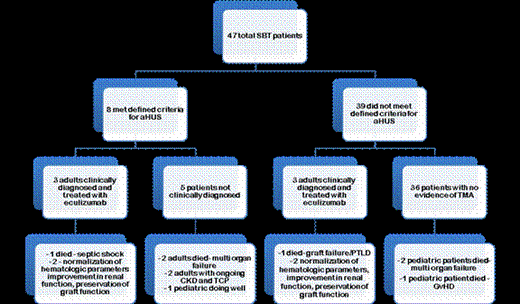
METHODS: This retrospective analysis was performed to evaluate whether we could identify a set of laboratory criteria that would reliably predict patients who are at higher risk of developing of aHUS in the pre-transplant or early post transplant period. Records of the forty-eight patients who underwent SBT at our institution between 2011 and 2013 were reviewed. Laboratory values were collected at three predetermined time points: pre-transplant (during transplant evaluation), 1-month post-transplant, and 6-12 months post-transplant. The criteria we designated for establishing a diagnosis of aHUS included fulfillment of all four of the following parameters at either the 1 month or 6-12 month time point: (1) 25% or greater decrease in platelet count from pre-transplant baseline, (2) serum creatinine increase of greater than 0.3 mg/dL from pre- transplant baseline, (3) haptoglobin of less than 50 mg/dL, and (4) LDH of greater than 250 units/L. Fisher exact test was used to find any statistical difference between the development of PT-aHUS and patient demographics and recorded cause of small bowel failure.
RESULTS: One patient was not included in the analysis due to immediate graft failure. Eight of the remaining 47 patients (17%) met the defined criteria for aHUS post transplant. Three of the 8 patients who met the defined aHUS criteria were diagnosed clinically with aHUS and were treated with eculizumab. The remaining 5 who met the defined laboratory criteria for aHUS but were not clinically diagnosed had the following outcomes: 2 patients died due to multi organ failure, 2 have ongoing chronic kidney disease (CKD) and TCP and 1 pediatric patient is doing well. Three patients who did not meet the defined laboratory criteria for aHUS were clinically diagnosed with aHUS and treated with eculizumab.
There were two significant pre transplant demographic associations that were identified by Fisher exact test with developing aHUS after SBT: age > 18 (RR 2.01, 95% CI 1.29-7.29, p=. 02) and ischemic bowel event preceding small bowel failure (RR 3.90, 95% CI 1.33-14.81, p=. 02).
There were two significant pre transplant demographic associations that were identified by Fisher exact test with developing aHUS after SBT: age > 18 (RR 2.01, 95% CI 1.29-7.29, p=. 02) and ischemic bowel event preceding small bowel failure (RR 3.90, 95% CI 1.33-14.81, p=. 02).
CONCLUSIONS: Utilizing the proposed criteria for defining PT- aHUS the incidence of PT- aHUS was 17% in our series, consistent with prior reports of PT- TMA in SBT patients. This incidence is higher than reported in renal transplant patients, suggesting some unique correlation between SBT and PT-aHUS. Multiple theories can be proposed for the etiology of complement dysregulation: (1) higher doses of immunosuppression (calcineurin inhibitor (CNI) based) required in small bowel recipients may augment endothelial damage and increase complement activity, (2) the transplanted organ itself may be more immunogenic and increase complement activity, (3) CNIs may deregulate T cell-complement homeostasis. The adult patients who met our criteria for PT-aHUS, but were not clinically recognized and treated had worse outcomes indicating a need for early diagnosis and treatment. The finding that an ischemic event as the etiology for small bowel failure is associated with an increased relative risk of developing post transplant aHUS is thought provoking and suggests that in some patients, bowel ischemia may actually be the initial TMA manifestation of aHUS. Pre-transplant evaluation of this subset of patients for the presence of known mutations in complement regulatory genes may be warranted.
Broome:Alexion Pharmaceuticals: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal