Abstract
Background: Heparin-induced thrombocytopenia (HIT) is associated with anti-platelet factor-4 (PF4)/heparin antibodies that activate platelets, resulting in thrombocytopenia and a pro-thrombotic state. At our institution antibody-mediated platelet activation is demonstrated by lumi-aggregometry, which is a method previously validated against the gold standard serotonin-release assay (SRA). Lumi-aggregometry does not involve radioactive isotopes, which is its major advantage over the SRA. The clinical course of HIT diagnosed via SRA and ELISA has been previously described, and clinical prediction tools such as the 4-T score were validated using these diagnostic tests. However, the clinical picture of HIT diagnosed by lumi-aggregometry has not been previously described.
Aims: The objective of this study is to describe the clinical and laboratory presentation of patients diagnosed with HIT by lumi-aggregometry.
Methods: Patients with clinically suspected HIT and quantitative anti-PF4 IgG-specific ELISA OD ≥0.400 (Gen-Probe, San Diego) received confirmatory HIT testing by lumi-aggregometry. Briefly, HIT antibody-induced activation of washed healthy donor platelets was tested at therapeutic (0.1U/mL and 0.5U/mL) and high (100U/mL) porcine heparin concentration. The degree of platelet activation was quantitated luminographically based on the light flash reaction of ATP (released from platelet dense-granules) with luciferin luciferase reagent. A ratio of therapeutic to high heparin luminescence amplitude of >5.0 and platelet aggregation at therapeutic, but not high, concentrations was considered a positive result. The results of assays performed by our regional HIT testing referral laboratory from June 2009 to July 2012 were reviewed to identify patients with positive HIT testing by lumi-aggregometry. Patient records were retrospectively reviewed to obtain predefined data on baseline patient characteristics, heparin exposure, platelet counts, and thrombotic events occurring in the 5 days preceding or the 30 days following the date of positive HIT testing.
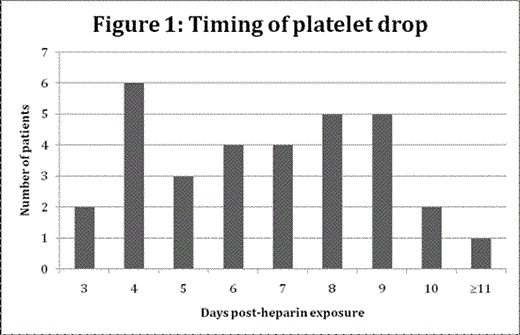
Results: We identified 43 patients diagnosed with HIT by lumi-aggregometry (median age 68.0, 49% male) while under the care of local academic (46%) or urban community hospitals (37.2% medical; 53.5% surgical; 9.3% intensive care). Median baseline platelet count was 187 (14-349). Median date of platelet drop post-heparin exposure was 6 days (range 3-14) in patients without prior heparin exposure or platelet transfusions (Figure 1). Platelet drop >50% and platelet nadir ≥20x109/L were present in the majority of patients (Table 1). Thrombocytopenia occurred prior to (70.5%) or the same day (23.5%) as thrombosis in 16/17 patients with serial platelet counts who developed HIT-associated thromboembolism.
Conclusion: Patients diagnosed with HIT by lumi-aggregometry present with similar findings to those described in SRA-confirmed HIT. These findings lend support to the use of lumi-aggregometry as an accurate diagnostic assay for the clinico-pathologic syndrome of HIT.
Percentage platelet drop from baseline and platelet nadir
| . | Percent platelet drop . | ||
|---|---|---|---|
| Platelet nadir | >50% | 30-50% | <30% |
| ≥20 x 109/L | 28 | 3 | 2 |
| 10-19 x 109/L | 5 | 1 | 0 |
| <10 x 109/L | 1 | 1 | 0 |
| . | Percent platelet drop . | ||
|---|---|---|---|
| Platelet nadir | >50% | 30-50% | <30% |
| ≥20 x 109/L | 28 | 3 | 2 |
| 10-19 x 109/L | 5 | 1 | 0 |
| <10 x 109/L | 1 | 1 | 0 |
Szkotak:Alexion Pharmaceuticals: Research Funding. Sandhu:Celgene: Honoraria; Jansen: Honoraria; Novartis: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal