Abstract
Background; Accelerated phase of chronic myeloid leukemia (AP-CML) is not clearly defined yet. There are different definitions to classify AP. In European Leukemia Net (ELN) 2013 recommendation, considerable therapeutic approach of de novo AP would be hematopoietic stem cell transplantation (HSCT) followed by frontline tyrosine kinase inhibitor (TKI). To explore long-term efficacy of frontline imatinib (IM) treatment and early predictors of long-term outcome, we analyzed a total of 73 patients who received frontline IM..
Method; AP defined as a definition of ELN recommendation.. A progression to blastic phase and loss of response were considered as progression. Patients who had received HCT were censored at the time of HCT when calculating overall survival (OS) and progression-free survival (PFS).
Results; Of 83 patients who diagnosed as AP, 73 patients received IM and other 10 patients had HSCT (n=7) or no treatment (n=3). Of 73 IM-treated patients, 36 patients maintained IM therapy and 37 patients discontinued IM with switch to 2G TKI (n=23) or HSCT (n=14). Analysis of baseline characteristics revealed prior cytogenetic response (CyR), and molecular response at 6 and 12 months for prediction of survivals.
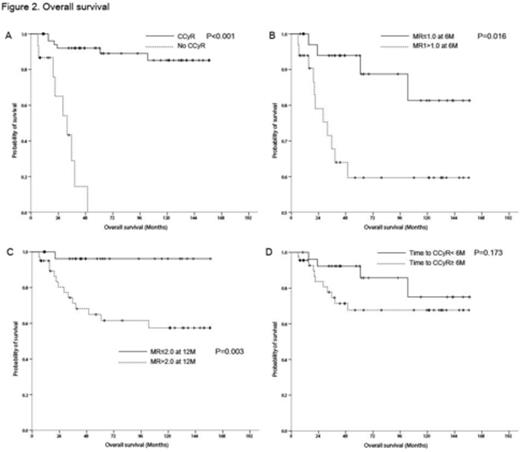
Clinical factors for better survival including Sokal score (p=0.203), Hasford sore (p=0.832), peripheral blood (PB) basophil count (p=0.374), spleen size (p=0.656), bone marrow (BM) promelocyte (p=0.839), BM basophil (p=0.478 were not significant. PB blast<10% (p=0.0670), PB eosinophil count>5% (p=0.031), platelet count >20x109/L (p=0.008), PB promyelocyte<2% (p=0.171), PB blast+promelocyte<20% (p=0.095), BM blast+promelocyte<20% (p=0.006), BM blast<10% (p=0.020) at diagnosis, achieving CCyR (p<0.001), achieving BCR-ABL PCRIS <10% (MR1.0) at 3M (p=0.020), BCR-ABL PCRIS <1% (MR2.0) at 6M (p=0.005) and MR2.0 at 12M (p=0.001) were included in multivariate analysis. Platelet count >20x109/L at diagnosis (p=0.002), achieving CCyR (p=0.007) and MR2.0 at 12M (p=0.048) were significant prognostic factors in multivariate analysis. Probability of 10Y OS for patients who acheived CCyR vs. no CCyR were 85.2% vs. 0% (p<0.001); median survival for patients without CCyR was 31.737 (95% CI, 16.269-47.147) months (Figure 2). Probabilities of 10Y OS for MR1.0 at 6M and MR2.0 at 12M were 81.3% vs. 59.7% (p=0.016) and 96.5% vs. 57.4% (p=0.003), respectively. However, time to CCyR<6M was not significant 10Y OS rate 75% vs. 67.6%, p=0.173). The 10Y PFS probability in patients who had acheived CCyR was 66.0% vs. 0% (p<0.001); median PFS for patients without CCyR was 9.462 (95% CI, 1.978-16.946) months. Probabilities of 10Y PFS in MR1.0 at 6M and MR2.0 at 12M were 63.2% vs. 44.9% (p=0.076) and 77.1% vs. 39.8% (p=0.005), respectively. Median PFS for patients not achieving MR1.0 at 6M and MR2.0 at 12M were 30.555 (95% CI, 0.0-61.252) and 22.867 (95% CI, 0.0-50.208), respectively. Time to CCyR<6M was not significant for PFS (10Y PFS rate 50.8% vs. 58.5%, p=0.828).
Conclusion: Achievement of CCyR or achievement of MR1.0 at 12M was important goals not only in progression but also in survival. Therefore if a patient doesnÕt achieve the goals, the treatments need to be changed.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal