Abstract
Shear stress (SS) can activate platelets, leading to exposure of surface P-selectin and development of platelet-leukocyte aggregates (PLAs). Increased numbers of PLAs has been reported in the circulation of patients with aortic valve stenosis (AS), who have high SS across the stenotic valves, and aortic valve replacement decreases the SS and number of PLAs. Activated platelets release their granule contents, which includes transforming growth factor-β1 (TGF-β1). Our lab has reported that SS can activate latent TGF-β1 released from platelets both in vitro and in in vivo mouse models of AS. Since current assays are not sensitive enough to detect activated TGF-β1 in plasma, we searched for evidence of active TGF-β1 in a mouse model of AS by assessing the level of phosphorylated Smad2/3, a downstream mediator of the classical TGF-β1 signaling pathway, in both circulating leukocytes free of platelets and PLAs.
We studied Ldlr-/-Apob100/100/Mttpfl/fl/Mx1Cre+/+ mice (Reversa), which spontaneously develop AS when fed a western diet (WD) (n=13), and compared them to the same mice fed a chow diet (n=13), who do not develop AS, and wild type (WT) mice (n=13). We identified leukocytes with an antibody to CD45 (clone 30-F11), platelets with an antibody to CD41 (clone MWReg30), PLAs by double staining, and p-Smad2/3 with an antibody to p-Smad2 (pS465/pS467)/Smad3 (pS423/pS425) (clone O72-670). Leukocytes and PLAs were analyzed by both a flow cytometer and an instrument that combines digital fluorescence microscopy with flow cytometry (ImageStream-X).
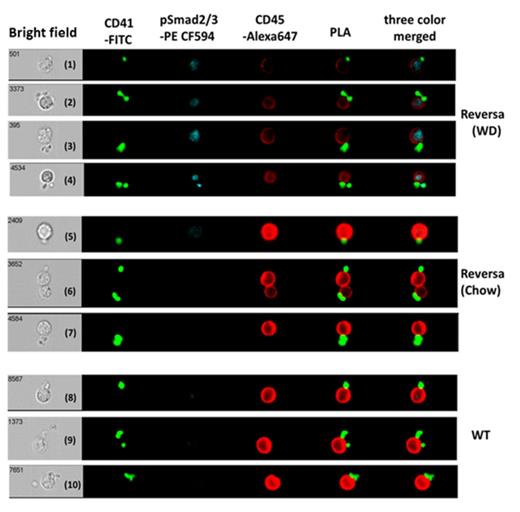
Reversa mice on WD for 3 months developed AS with increased SS across the valves and increased plasma total TGF-β1 levels in circulation as we previously reported (Wang et al., ATVB, 2014). Reversa mice on WD also had a significantly increased percentage of PLAs compared to the other two groups (both p<0.05; Table), suggesting platelet activation under high SS. The intensity of p-Smad2/3 staining in leukocytes free of platelets of Reversa mice on a WD was greater than the intensity in the other two groups (both p<0.01). In all 3 groups, p-Smad2/3 in PLAs was higher than in leukocytes free of platelets (all p<0.001), but Reversa mice on a WD had the highest p-Smad2/3 levels in their PLAs (p<0.05 for both. Fluorescent microscopy showed that p-Smad2/3 was located primarily in the leukocytes of the PLAs (Figure 1). There was no correlation between p-Smad2/3 staining of PLAs and the number of platelets per leukocyte (p=0.78 for WT group, p=0.52 for Reversa chow diet, and p=0.15 for Reversa WD).
In summary: (1) PLAs have more leukocyte p-Smad2/3 than leukocytes that are free of platelets, regardless of genotype/diet. (2) Leukocytes free of platelets in Reversa mice on a WD have more p-Smad2/3 than those in the other two groups. (3) Reversa mice on WD have both more PLAs and more leukocyte p-Smad2/3 in their PLAs. We conclude that the increased leukocyte p-Smad2/3 in Reversa mice on WD reflects increased TGF-β1 signaling through both: 1) direct platelet-leukocyte interactions and 2) release of TGF-β1 from platelets into plasma and its subsequent activation by high SS.
| Group . | PLA (% of total leukocytes) . | p-Smad2/3 Mean Fluorescence Intensity (AU) . | |
|---|---|---|---|
| Leukocytes free of platelets . | PLAs . | ||
| WT | 16.9 ± 1.9 | 173.9 ± 15.49 | 204.7 ± 17.9 # |
| Reversa (chow diet) | 16.4 ± 2.1 | 172.3 ± 15.24 | 200.8 ± 15.9 # |
| Reversa (WD) | 26.1 ± 3.1*^ | 237.5 ± 17.42*^ | 268.9 ± 17.1 *^# |
| Group . | PLA (% of total leukocytes) . | p-Smad2/3 Mean Fluorescence Intensity (AU) . | |
|---|---|---|---|
| Leukocytes free of platelets . | PLAs . | ||
| WT | 16.9 ± 1.9 | 173.9 ± 15.49 | 204.7 ± 17.9 # |
| Reversa (chow diet) | 16.4 ± 2.1 | 172.3 ± 15.24 | 200.8 ± 15.9 # |
| Reversa (WD) | 26.1 ± 3.1*^ | 237.5 ± 17.42*^ | 268.9 ± 17.1 *^# |
* p<0.05 Reversa (WD) vs. WT; ^ p<0.05 Reversa (WD) vs. Reversa (chow diet); # p<0.001 PLA vs. leukocytes free of platelets. AU: arbitrary unit; WD: western diet
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal