Abstract
BACKGROUND: HMG-CoA reducatase inhibitors, or statins, are commonly prescribed medications which improve life expectancy in general population. They are known to improve hypercholesterolemia and decrease the incidence of cardiovascular events including myocardial infarction or stroke, but also suggested for cancer prevention even though the mechanism is not fully elucidated. Recent studies emphasized the potential role of statins in the cancer treatment to increase response rate to chemotherapy and to improve survival of cancer patients. Statin family of drugs is known to trigger tumor specific apoptosis and to result in growth arrest in leukemias (Penn, Leukemia 2002).
The promising result of STIM (STop Imatinb) trial suggested that successful discontinuation of tyrosine kinase inhibitor (TKI) in patients with chronic myeloid leukemia (CML) is possible when patients attain deeper molecular response (defined as 4.5 log reduction or deeper) for 2 years or longer. However, with our current knowledge, there is no known additive intervention facilitates the achievement of MR4.5. We hypothesized that the use of statin improves response rate to tyrosine kinase inhibitor (TKI) therapy in patients with chronic myeloid leukemia (CML), thus increasing MR4.5 achievement and increasing the chance of being attempted TKI discontinuation.
METHODS: A total of 503 patients treated with TKI for CML treatment were initially evaluated for the response to TKI therapy with respect to complete cytogenetic response (CCyR), major molecular response (MMR) and molecular response at 4.5 (MR4.5) and statin use. The inclusion criteria confined the patients with chronic phase (CP) treated with imatinib at the dose of 400mg daily, thus excluding 95 patients. Finally, 408 patients were entered into the final analysis. The statin group was defined as those on statin for cholesterol control at the time of imatinib commencement and remaining on statin while on at least 3 years or longer. Cumulative incidence method considering competing risk was adopted to calculate the incidences of MCyR, CCyR, MMR and MR4.5. Discontinuation of imatinib was accounted as competing risk in the analysis. Treatment failure, progression free- and overall survival was also evaluated.
RESULTS: With a median follow-up duration of 6 years (range 3 months to 14 years), 88 patients (21.3%) were defined as statin group. Types of statin includes atorvastatin (n=44, 50%), rosuvastatin (n=26, 30%), simvastatin (n=10, 11%), pravastatin (n=6, 7%) and fluvastatin (n=2, 2%).
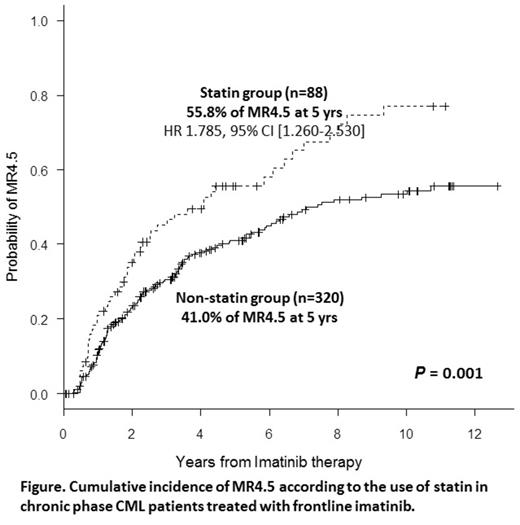
The MCyR and CCyR achievement was not significantly different between the 2 groups (p=0.769 for MCyR and p=0.091 for CCyR). No difference of CCyR at 12 months was noted: 70.1% in statin vs 62.8% in non-statin group. The statin group showed a higher response rate than non-statin group for MMR (p=0.005) and MR4.5 (p=0.001): 67.3% vs 49.2% for MMR at 18 months; 55.8% vs 41.0% for MR4.5 at 5 years (Figure).
Multivariate analysis was successful to confirm the use of statin as an independent clinical factor for improving MR4.5 (HR 1.785, 95% CI [1.260-2.530], p=0.001), but other clinical factors were not identified such as Sokal risk, age, gender or additional cytogenetic abnormalities (ACAs) at presentation. For MMR, the use of statin was also confirmed as independent factor for MMR (HR 1.541 95% [1.015-2.341], p=0.043) in addition to ACAs (HR 0.381, p=0.0038) and high sokal risk (HR 0.687, p=0.042).
The use of statin was not found to be associated with improvement in treatment failure (p=0.580), progression free survival (p=0.731) or overall survival (p=0.542) in the present study.
CONCLUSION: The use of statin suggested to improve deeper molecular response following imatinib therapy in CML-CP patients, therefore is promising to increase chance of attempt to TKI discontinuation. Statin appears to improve the chance of MR4.5 achievement by 78.5%. Careful interpretation is required and replication study in an independent cohort is strongly warranted to reach a clear conclusion on this promising effect of statin. The use of statin to deepen the molecular response of TKI therapy should be evaluated in the context of clinical trial.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal