Abstract
Background
Partial tandem duplication mutations of the Mixed Lineage Leukemia gene (MLL-PTD) can be found in about 10% of patients with AML, especially in patients with normal karyotype AML. The mutation generates a self-fusion within the N-terminal part of MLL and has been shown to be leukemogenic in mouse models. In patients, the presence of the mutation is associated with poor prognosis. Little is known on the molecular profile of patients with MLL-PTD and on the cooperating mutations. In order to identify accompanying molecular alterations, we performed whole exome sequencing (WES) of eight AML patients harbouring MLL-PTD mutations. Based on the observed alterations we then designed a custom amplicon panel and performed targeted resequencing in a cohort of 90 MLL-PTD mutated AML patients.
Materials and Methods
All patients included in this analysis were treated in prospective treatment protocols of the Study Alliance Leukemia (SAL). To enrich for malignant cells and to obtain germline reference material (T-cells), FACS sorting was performed on viable cells banked at diagnosis. After whole genome amplification of the primary DNA, whole exomes were enriched (TruSeq chemistry; Illumina), and paired-end sequenced using Illumina HiSeq2000 2x100 bp runs. Resulting data were mapped against human genome (Hg19). Only somatic single nucleotide variants (SNVs) were included in the final analysis. Based on the SNVs identified by whole exome sequencing (WES), a custom amplicon panel (TruSeq Custom Amplicon, TSCA, Illumina) for targeted resequencing was designed. The assay included either the entire coding region or mutational hot spots of 56 genes (Fig.1). In total, 700 targets were amplified in a single reaction for each patient and paired end sequenced on a MiSeq NGS system (Illumina). Paired end reads were BWA mapped against targeted regions and data analysis was done using the Sequence Pilot software package (JSI Medical Systems) with a 20% variant allele frequency (VAF) mutation calling cutoff. Only non-synonymous variants not specified as SNP in the db137 database and predicted as deleterious (Provean) were included in the final analysis. All variations were confirmed by Sanger sequencing.
Results
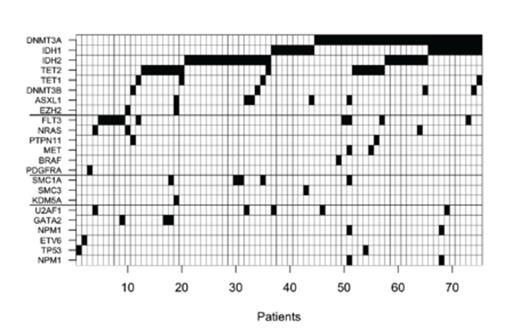
WES of eight MLL-PTD (7/8 FLT3-ITD negativ) patients revealed a total 490 SNVs (range 13-254 per patient). Most frequently mutated genes were DNMT3A, IDH1/2 and TET2. Somatic mutations were also found in genes rarely mutated in AML, such as ATM, GNAS, TET1 and EP300. Based on the WES-data, 90 MLL-PTD patients were screend for a panel of 56 genes using the TSCA assay, which revealed in total of 169 mutations. 18 genes were not found to be mutated and in 8 patients, no co-occurring mutations were identified. Due bad assay performance EP300, EZH1, JAK3, MLL2, MLL3 and NOTCH1 were excluded from the data analysis. Here again, the most frequently mutated genes were DNMT3A (34.4%), IDH1 (20.0%), IDH2R140 (18.9%), IDH2R172 (7.9%), TET2 (16.7%) and FLT3 (11.3%). Mutations were less frequently found in RUNX1 (8.9%) and ASXL1, SMC1A, U2AF1 (5.6% each) (Fig. 1). In addition to these known genes, most prevalent mutations were found in ATM (8.9%) as well as DNMT3B and TET1 (4.4% each). Overall, we oberserved a low frequency of mutations in typical class 1 genes such as NRAS, KRAS and FLT3, which was lower than reported in the TCGA data set.
Conclusions
This analysis in a large set of patients with MLL-PTD mutations did not reveal any new and specific individual mutation present in patients with this alteration. Instead, our finding of a very high prevalence of alterations in epigenetic regulator genes, found in more than 85% of patients with MLL-PTD, strongly argues for a particular disease biology in these patients. These findings might also implicate that treatment based on demethylating agents or histone-deacetylase inhibitors might be especially attractive in patients with MLL-PTD.
The assay included either the entire coding region or mutational hot spots of the following 56 genes; ASXL1, ATM, BCOR, BRAF, CBL, DDR1, DNMT1, DNMT3A, DNMT3B, EIF4A2, EP300, ETV6, EZH1, EZH2, FLT3, GATA1, GATA2, GNAS, HRAS, IDH1, IDH2, JAK1, JAK2, JAK3, KDM4A, KDM5A, KDM5C, KDM6A, KIT, KRAS, MET, MLL, MLL2, MLL3, NOTCH1, NOTCH4, NPM1, NRAS, PDGFRA, PDGFRB, PHF6, PTEN, PTPN11, RAD21, RUNX1, SF3A1, SF3B4, SMC1A, SMC3, SMC4, TET1, TET2, TP53, U2AF1 and WT1.
Thiede:AgenDix GmbH: Equity Ownership, Research Funding; Illumina: Research Support, Research Support Other.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal