Abstract
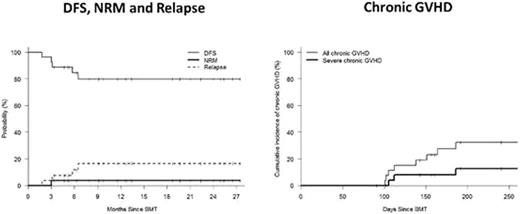
Calcineurin inhibitors (CNIs) form the foundation of current GVHD prophylaxis regimens. Although advances in immunosuppressive regimens have had a significant impact on the incidence and severity of acute GVHD, it is noteworthy that CNIs have had little impact on chronic GVHD. We hypothesized that a CNI-free regimen consisting of post-transplant cyclophosphamide (Cy) and brief-course sirolimus would decrease the risk of chronic GVHD and non-relapse mortality and improve outcomes following reduced intensity allogeneic PBSCT. Twenty-seven patients with high risk hematologic malignancies were enrolled in the study: median age 61 years (25-73). All patients had a 10/10 locus matched donor; MRD=18, MUD=9. Conditioning consisted of fludarabine 30mg/m2 on days -9 to -6, IV busulfan 130 mg/m2 on days -5 to -4, and Cy 14.5 mg/kg on days -3 and -2 followed by unmanipulated PBSCT. Post-grafting immunosuppression consisted of Cy 50 mg/kg/day on days 3 and 4 and sirolimus starting day +5 and completing day+90 in the absence of GVHD. Donor engraftment occurred in all patients with a median time to neutrophil and platelet recovery of 15 and 30 days, respectively. The median day +90 donor T cell and myeloid chimerism was 94% (40-100%) and 100% (11-100%) respectively. Three patients received donor lymphocyte infusions for incomplete donor T cell chimerism. The cumulative incidence of grade II-IV acute GVHD, grade III-IV acute GVHD, all chronic GVHD, and severe chronic GVHD was 41%, 15%, and 32%, and 12% respectively. Non-relapse mortality (NRM) and relapse incidence at 2 years was 4% and 17% respectively. With a median follow-up of 18 months, the estimated 2 year overall and disease-free survival was 71% and 80% respectively for the whole cohort, while it was 87% and 89%, respectively in the subgroup of 18 patients receiving MRD transplants. Good immune reconstitution was evidenced by low cytomegalovirus reactivation rates, occurring in only 4 of 19 at-risk patients (21%). Transplant-related toxicity included BK virus-associated cystitis in 33% of patients and a non-fatal hepatotoxicity syndrome in three patients consisting of transaminase elevation and ascites, with resolution following discontinuation of sirolimus. CNI-free GVHD prophylaxis with post-transplant Cy and brief-course sirolimus achieves consistent donor engraftment, low rates of GVHD and NRM, and excellent outcomes in recipients of HLA-identical donor allogeneic PBSCT.
Off Label Use:fludarabine, cyclophosphamide, and sirolimus are not FDA-labeled for stem cell transplantation.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal