Abstract

Impaired cognition – an increasingly recognized concern after HCT – has significant potential to impact societal reintegration. Previous studies have focused on recipients of full-intensity allogeneic HCT; the trajectory of cognitive function after reduced-intensity HCT is unclear. Furthermore, the pathogenesis of cognitive impairment after HCT is unknown. Telomeres are repetitive DNA-protein structures localized to chromosome ends that protect chromosome integrity. Telomeric shortening occurs with each cell division; chemo/radiation hastens telomeric attrition. Glial cells are mitotic and susceptible to telomeric shortening. Constitutional telomere length using blood DNA is representative of the whole organism. Telomeric shortening (measured in blood) is associated with Alzheimer's disease severity. Shorter telomeres could play a role in cognitive impairment after HCT.
The longitudinal trajectory of cognitive function was assessed from pre-HCT (n=194), to 6m (n=165), 1y (n=155), 2y (n=125) post-HCT using standardized neuropsychological tests (14 tests assessing 8 domains: executive function, processing speed, verbal speed, verbal fluency, working memory, auditory memory, visual memory, fine motor dexterity). IQ was assessed to estimate cognitive reserve. Cognitive function was also assessed in age-, gender-matched healthy controls (HC: n=98) at corresponding time points. Generalized estimating equations (GEE) were fitted to transformed HC scores using time, IQ and sex as covariates; these were then used to compute fitted scores and residuals (fitted – observed scores) in HCT recipients, thus ensuring that HCT residuals were devoid of practice effects. Standardized HCT residuals were transformed to T-scores (mean=50 and SD=10) for GEE analysis. Blood germline DNA was procured pre-HCT (n=142) to assess relative telomere length (RTL: ratio of telomeres to single genes) using qPCR-based telomere assay. RTL was dichotomized as short vs. long (< vs. ≥ median). p≤0.01 was used as critical value to account for multiple comparisons.
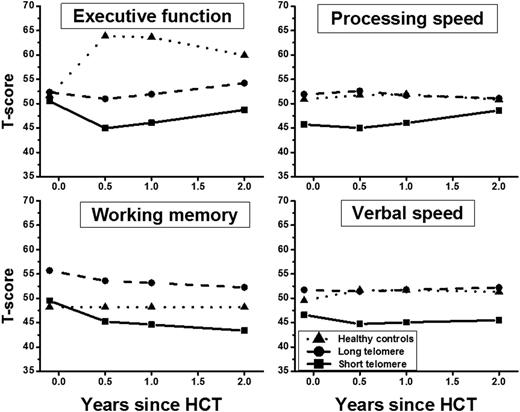
Median age at study was 49y for HCT recipients (range: 19-71) and 51y for HCs; primary diagnoses included acute leukemia (69%), lymphoma (14%) and others (17%). Reduced-intensity conditioning was used in 53% of HCT recipients. Myeloablative total body irradiation was used in 72% of full-intensity HCT recipients. Fifty-one percent of all HCT recipients developed chronic graft-versus-host disease. Compared with HCs, HCT recipients as a whole demonstrated lower cognitive function post-HCT in executive function, p=0.0008; processing speed, p=0.003; verbal fluency, p=0.003; motor dexterity, p=0.001. Multivariable longitudinal analysis of HCT recipients identified older age, male gender, Hispanic ethnicity, low education, income and cognitive reserve, high risk of relapse and high fatigue as significant contributors to cognitive impairment after HCT. After adjusting for these variables, cognitive function was worse in patients who received full-intensity HCT (compared with reduced-intensity) conditioning in executive functioning, p=0.01; processing speed, p=0.0005; verbal speed, p<0.0001; visual memory, p=0.002 (Fig 1). Importantly, there were no significant differences in cognitive functioning between reduced-intensity HCT recipients and HCs (p>0.1; Fig 1). Longitudinal multivariable analysis showed a significant association between short telomeres measured prior to HCT and post-HCT cognitive reduction in female HCT recipients for executive function, p=0.004; processing speed, p=0.009; verbal speed, p=0.009; and working memory, p=0.003 (Fig 2).
We demonstrate several new findings in this study: patients receiving full-intensity HCT are at risk for cognitive impairment in executive functioning, processing speed, verbal speed and visual memory; those receiving reduced-intensity HCT are generally spared. In addition, telomeric shortening prior to HCT is associated with poorer executive function, processing speed, verbal speed and working memory in females after HCT, and not males. Identifying vulnerable subpopulations will facilitate implementation of prevention strategies.
Fitted longitudinal T-scores for full- and reduced-intensity HCT patients and HCs
Fitted longitudinal T-scores for full- and reduced-intensity HCT patients and HCs
Fitted longitudinal T-scores for female patients with short and long telomeres and HCs
Fitted longitudinal T-scores for female patients with short and long telomeres and HCs
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal