Abstract
In Epstein-Barr virus (EBV) classical Hodgkin lymphoma (EBV+ cHL), Hodgkin-Reed Sternberg cell antigen presentation is intact, with viral expression restricted to sub-dominant latent-antigens including LMP1/2A. Large epidemiological studies have reported differential HLA-class I (HLA-I) susceptibility to EBV+ cHL. The functional basis for these observations is unknown. HLA-I molecules present viral peptides for recognition by CD8+ T-cells, and it may be that the relative risk of developing EBV+ cHL is due to HLA-I alleles influencing the magnitude of CD8+ T-cell immunity against relevant EBV-specific antigens. However this remains speculative, with immunological evidence lacking. Several non-HLA-I linked genetic susceptibility loci have been identified, and HLA-I associations may simply represent markers for genes of diverse functions that are in linkage disequilibrium to the HLA-I region. We undertook an Australasian Leukaemia and Lymphoma Group study to address this fundamental question, utilizing 4 distinct but complimentary experimental approaches.
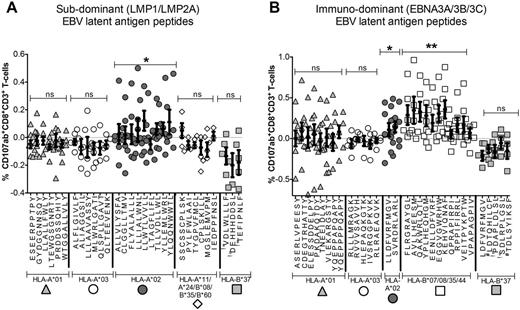
1. 9 EBV+ cHL and 11 EBV-ve cHL pre-therapy PBMC samples were tested for ex-vivo IFNγ, TNFα and CD107a CD8+ T-cell immunity, using overlapping LMP1 and LMP2A peptide pools. The non-HRS expressed EBV-lytic protein BZLF1 was a control. Highly stringent FACS gating was used to maximize specificity. Results were interrogated using Profile and SPICE analysis. Interestingly IFNγ, TNFα and CD107 CD8+ T-cell responses in HLA-A*02 EBV+ cHL (but not EBV-ve cHL) patients were greater than non-HLA-A*02 (LMP1 p=0.002; LMP2A p=0.03; combined LMP1/LMP2A p=0.005), whereas BZLF1 was equivalent, indicating that HLA-I provides differential CD8+ T-cell immunity against relevant EBV-latent antigens in EBV+ cHL but not EBV-ve cHL.
2. However, up to 4 different HLA-A/B molecules can potentially present relevant EBV-derived epitopes in each individual, adding a confounding layer of complexity to single allele-based effects. To overcome this and enhance sensitivity, we used the mutant HLA-I 721.221 cell-line (pulsed with LMP2A), transfected with either HLA-A*01, HLA-A*02, HLA-A*03 or HLA-B*08 alleles, as antigen presenting cells to in-vitro expand LMP2A-specific CD8+ T-cells from HLA-A*02 heterozygotes. This found ∼90% of the HLA-I LMP2A response was restricted through HLA-A*02.
3. In contrast to EBV+ cHL, in EBV-post-transplant lymphoproliferative disorders (EBV+ PTLD) the immunogenic EBNA3A/3B/3C latent-antigens are expressed. We compared HLA-I associations in 110 cHL (35% EBV+ cHL) to 153 PTLD (63% EBV+ PTLD) patients. Using Bonferoni corrected statistics, we confirmed that HLA-A*02 and HLA-A*01 homozygotes had lower and higher susceptibility to EBV+ cHL respectively, and that HLA-B*37 was positively associated. Notably, no HLA-I associations with EBV+ PTLD were found.
These data combined illustrate that differential HLA-I-associated susceptibility to EBV+ cHL reflects altered EBV latent antigen-specific CD8+ T-cell immune hierarchies. For lymphomas expressing a restricted set of poorly immunogenic proteins, even modest CD8+ T-cell responses against relevant tumor-associated proteins confer protection, with broad implications for EBV-vaccine design. Studies are required to determine if similar mechanisms are applicable to non-lymphoid EBV+ malignancies with restricted latency such as undifferentiated nasopharngeal carcinoma.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal