Abstract
Immuno-chemotherapy with Rituximab and CHOP (R-CHOP) represents the standard of care for Diffuse Large B Cell Lymphoma DLBCL patients of any age. The clinical outcome of patients treated by this program has been reproducibly confirmed by many independent randomized clinical trials that recruited patients on the basis of restrictive inclusion/exclusion criteria. Few information is available about the short and long term outcome of patients who, in the daily clinical practice, are treated by R-CHOP despite the presence of one or more co-morbidities that should have been considered as exclusion criteria for the aforementioned clinical trials.
To evaluate the response rate, the early death as well as overall survival of DLBCL patients who received a front-line R-CHOP or R-CHOP/like programs despite the presence at diagnosis of one or more severe comorbidities.
From April 2000 to March 2013 we evaluated 473 consecutive and unselected DLBCL homogeneously treated at our institution with R-CHOP or R-CHOP/like as first line treatment. These patients were males in 53%, had a median age of 64 years (range 18 -89), clinical stage III-IV in 54%, B symptoms in 31%, bone marrow involvement in 14%, Bulky disease (> 7 cm) in 34%, Extranodal presentation (>1 site) in 30%, ECOG (>2) in 25%, a high LDH level in 48% and high IPI (>2) in 42%. For the present analysis, patients were divided in group A (n=257) and B (n=216) according to whether they met or not clinical trial enrollment criteria respectively (as reported by Cunningham et al, Lancet 2013). Reasons for not meeting these criteria were the following organ dysfunctions: cardiovascular (75%), liver (35%), lung (14 %) and renal (5%) or the presence of a positive serology for HCV, HBV and HIV that were detected in 73%, 38% of and 10% of these patients, respectively. Of note, 41 patients (8%) had a previous solid tumor. Overall 63% of patients had only one comorbidity, 30% more than two and 6% more than three.
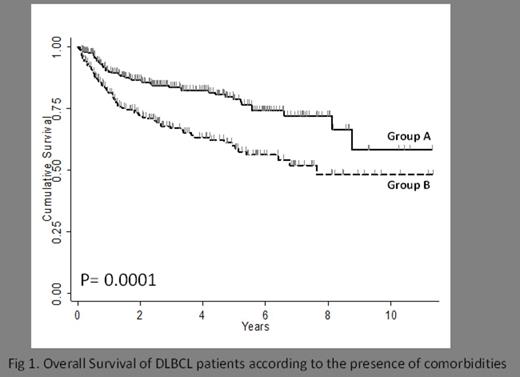
Gender and patients with bulky disease were equally distributed between group A and B. However, patients belonging to group A were younger (58 vs 70, p=0.0001), had a less advanced stage disease (stage III and IV 46% vs 54, p= 0,001), fewer B symptoms (45% vs 55%, p= 0,024), less BM infiltration (34% vs 65%, p= 0,002), less extranodal involvement (42% vs 57%, p= 0,004) and showed a better performance status (ECOG >2 41% vs 59%, p= 0,005), a lower proportion of cases with high LDH level (46% vs 54%, p= 0,002) and a better IPI (IPI >2, 40% vs 60%, p= 0,000). After 6 chemotherapy cycles, the response rate was slightly lower in group B (82% vs 89%, p=ns). The proportion of early deaths (events registered within 6 months from start of chemotherapy), was lower in group A (7/257, 2.7%) as compared to group B (22/216, 10.2%, p=0.001), independently of age (> or<60 year). Of note, the early death rate in group A reproduced that seen in several randomized clinical trials. Main reasons of early death were: bacterial infections, acute liver failure, myocardial infarction and progressive disease. The median time to early death was 1.8 months in group A as compared to 2.35 in group B (p= ns), respectively. By multivariate logistic regression analysis, corrected by age, IPI and bone marrow positivity, the presence of B symptoms (OR 3.32, 95% CI 1.23-9.01, p= 0.018) and the presence of any comorbidity (OR 3.13, 95% CI 1.07-9.14, p= 0.037) proved as strongly independent predictors of early deaths. The 2 year probability of overall survival was 87% and 72% (p= 0.0001) in group A and B, respectively (Figure 1). B symptoms, and the presence of any comorbidity and IPI >2 were also significantly affecting the long term overall survival (B symptoms, HR 1.59, 95% CI 1.04-2.43; any comorbidity, HR 1.71, 95% CI 1.12-2.61; IPI >2, HR 2.38, 95% CI 1.48-3.82). Based on this analysis, we finally identified three risk categories: low (no B symptoms, no comorbidity and no high IPI), intermediate (presence of either B symptoms or any comorbidity or high IPI) and high (co-presence of B symptoms, any comorbidity and High IPI) that were associated to a significantly different 2 year survival of 93%, 81% and 48% (p= 0.0000), respectively.
The clinical outcome of a large proportion of DLBCL patients treated in the daily clinical practice is negatively affected by comorbidities. This should be considered when analyzing transferability of results from randomized clinical trials into clinical practice.
Rambaldi:Roche, SpA, Italy: Consultancy, Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal