Abstract
Despite the remarkable improvement in survival of children with acute leukemia in the last decades, some patients still have a poor outcome. FMS-like tyrosine Kinase 3 (FLT3) is a tyrosine-kinase receptor with a key role in hematopoiesis whose mutations and overexpression have emerged as negative prognostic biomarkers in childhood leukemia. Infant leukemias (those diagnosed at age<1 year) are a special subgroup characterized by distinctive clinico-biological features, including frequent MLL (Mixed Lineage Leukemia) gene rearrangements, high FLT3 expression and high sensitivity to cytarabine (Ara-C), but a dismal prognosis. Ara-C is a deoxycytidine analog known to be transported by the human equilibrative nucleoside transporter 1 (hENT1) across the cell membrane. Once inside the cell Ara-C is activated by deoxycitidine kinase (DCK) to finally exert its cytotoxic action. High hENT1 expression levels have been involved in Ara-C sensitivity in patients with acute leukemia. However, the mechanisms that regulate the expression of hENT1 and its activity, as well as the putative relationship between FLT3 and Ara-C transport and metabolism are scarcely known.
To study the role of FLT3 in the regulation of the expression and activity of the main Ara-C transporters and metabolizing enzymes (ME) in pediatric leukemia.
Bone marrow samples of 56 pediatric patients diagnosed with acute leukemia in Hospital Sant Joan de Déu and 3 acute leukemia cell lines (MV4-11, SEM, K562) were used for screening of Ara-C transporters and ME expression. In all cases the patients or their parents signed an informed consent. We performed a positive selection of cases with acute lymphoblastic leukemia (ALL) with hyperdiploidy (>50 chromosomes) and of MLL-rearranged cases presenting either as ALL or acute myeloblastic leukemia (AML), as these are the subgroups with highest expression of FLT3 reported in the literature, although other leukemia subtypes were also represented. The mRNA expression levels of FLT3, nucleoside transporters (NT) (hENT1, hENT2, hCNT1, hCNT3) and Ara-C ME, dCK and CNII, were quantified using real-time PCR and analyzed with the 2-ΔΔCt method, with non-neoplastic samples used as controls for the relative quantification. Direct nucleoside and Ara-C uptake measurements were performed using [5,6-3H]-nucleosides. The role FLT3 might play in the expression of NT and ME genes as well as on the activity of Ara-C transporters in these cell lines was addressed by repressing FLT3 function with its specific inhibitor PKC412 (sold only for research purposes).
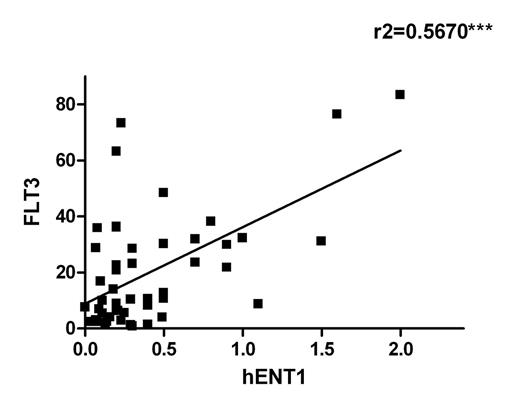
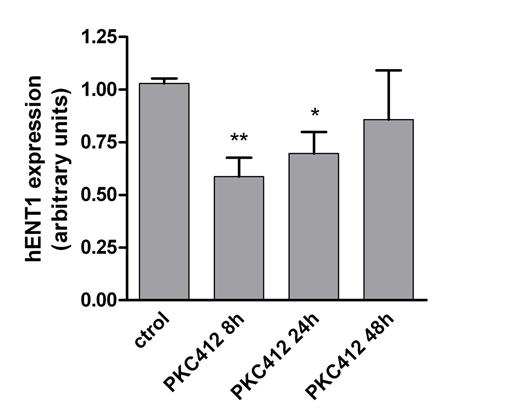
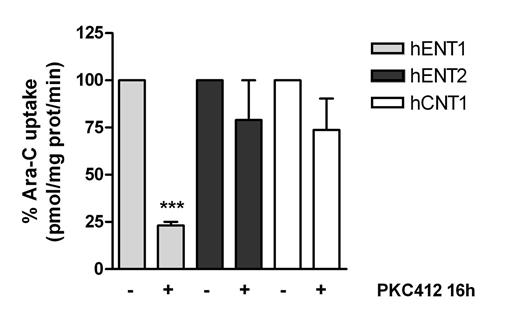
We included 56 patients (68% males) diagnosed with acute leukemia. The median age was 5.3 years (range 0-16.2), with 3 cases of infant leukemia. Fifty cases (89%) were precursor B-ALL (24 hyperdiploid cases, 5 MLL rearranged, 3 BCR-ABL+, 4 E2A-PBX1+, 5 TEL-AML1+, 9 other subtypes), 5 cases were AML (4 MLL positive cases and one case with mutated FLT3) and one case was a T-ALL harboring FLT3 mutation. As expected, the FLT3 expression was higher in cases with ALL and MLL rearrangement and, to a less extent, in ALL with hyperdiploidy. Interestingly, a significant positive correlation was found between FLT3 and hENT1 expression (mRNA levels) with all patient samples (figure 1). hENT1 expression and cytarabine-mediated uptake was significantly repressed when MV4-11 and SEM cell lines were exposed to the FLT3 inhibitor PKC412 (figures 2 & 3) without affecting hENT2, CNT1 and CNT3 expression and activity.
Our results show a strong correlation between FLT3 and the Ara-C transporter hENT1 in pediatric leukemia patients. This observation was consistent with the in vitro evidence that FLT3 inhibition resulted in hENT1 repression and down-regulation of Ara-C uptake in leukemic derived cell lines. Taken together, our data suggest that FLT3 regulates hENT1, thereby modulating the ability of cancer cells to incorporate Ara-C and promote its cytotoxic action. As FLT3 inhibitors are currently being tested as mono or combined therapy with Ara-C in several clinical trials, based upon our observations we suggest that a better schedule design might eventually be needed when dealing with treatments involving FLT3 inhibitors and Ara-C, thereby improving the outcome of this subset of patients.
Off Label Use: The presentation include the results of in vitro studies with the FLT3 inhibitor PKC412. This drug was only used for in vitro studies, exclusively for research purposes.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal