Abstract
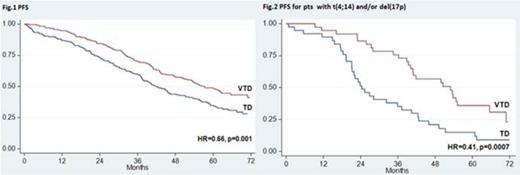
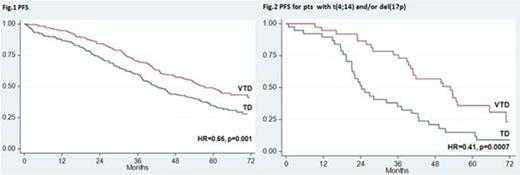
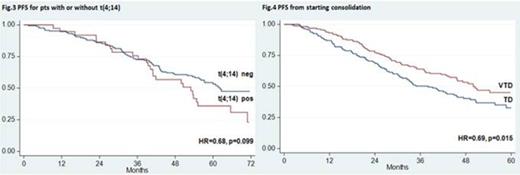
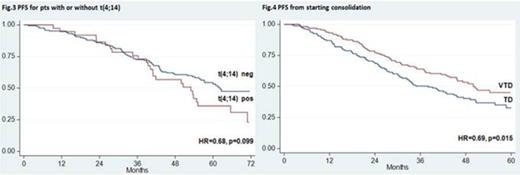
Over the last years, incorporation of novel agents into autologous stem cell transplantation (ASCT) has improved markedly the outcomes of younger patients with newly diagnosed multiple myeloma (MM). Superior results with experimental treatments vs previous standards of care have been frequently reported after preliminary analyses and need to be confirmed with longer follow up. The randomized phase 3 GIMEMA-MMY-3006 study was designed to compare bortezomib-thalidomide-dexamethasone (VTD) vs thalidomide-dexamethasone (TD) as induction therapy before, and consolidation after, double ASCT. Data from the initial analysis, with a median follow up of 36 months, demonstrated that patients randomized to the VTD arm enjoyed superior complete/near complete response (CR/nCR) rates after both induction and consolidation therapy, and had a significantly longer PFS compared to those prospectively assigned to the TD arm. We performed an updated analysis of the study after a median follow up of 59 months and results are herein reported. A persistent TTP and PFS benefit with incorporation of VTD into ASCT was confirmed. On an intention-to-treat analysis of 236 patients randomized to the VTD arm, median TTP was 62 months and median PFS was 57 months. The median values for 238 patients randomly assigned to the TD arm were 45 months for TTP (HR=0.64, p=0.001) and 42 months for PFS (HR=0.66, p=0.001) (Fig. 1). With the longer follow up of this analysis, an initial divergence between OS curves could be appreciated after 4 years, although the difference was not yet statistically significant at 6 years (75% for VTD vs 69% for TD). Superiority of VTD over TD for TTP and PFS was retained across prespecified subgroups of patients with high risk and low risk disease. In particular, PFS benefit with VTD was seen for patients age >60 years (HR=0.62, p=0.013) and younger than 60 years (HR=0.70, p=0.026), with ISS stage 1 (HR=0.59, p=0.009) and ISS stage 2-3 (HR=0.69, p=0.018), and for those with t(4;14) and/or del(17p) (HR=0.43, p<0.001) and with t(4;14) alone [t(4;14) positivity but lack of del(17p)] (HR=0.41, p=0.001). In comparison with patients with t(4;14) positivity who were randomized to TD, those assigned to the VTD arm had significantly longer PFS (median: 24 vs 53 months, HR=0.41, p=0.0007) (Fig. 2) and a trend towards longer OS (4-year estimates: 66% vs 81%, p=0.052). By the opposite, similar PFS curves were seen for patients in the VTD group regardless of the presence or absence of t(4;14) (Fig. 3). On multivariate Cox regression analysis, randomization to the VTD arm was an independent factor predicting for prolonged PFS (HR=0.64, P=0.001). Additional disease- and treatment-related variables independently affecting PFS included attainment of CR/nCR after both induction (HR=0.64, p=0.010) and consolidation therapy (HR=0.57, p<0.001), β2-m >3.5 mg/L (HR=1.7, p<0.001) and presence of t(4;14) and/or del(17p) (HR=2.0, p<0.001). On multivariate analysis, β2-m, cytogenetic abnormalities and attainment of CR/nCR after consolidation therapy were independently associated with OS. With an updated median follow-up of 49 months from the landmark of starting consolidation therapy, median PFS was 50 months for patients receiving VTD consolidation and 38 months for those treated with TD (HR= 0.69, P=0.015) (Fig. 4). Superior PFS with VTD vs TD consolidation therapy was observed for patients who failed CR/nCR after the second ASCT (HR=0.48, P=0.003) and was retained in both low risk and high risk subgroups. Finally, duration of OS from relapse or progression was similar between the two treatment groups (median, 42 for VTD vs 35 months for TD, p=0.47), even when bortezomib was incorporated into salvage therapy. In conclusion, this updated analysis of the GIMEMA-MMY-3006 study demonstrated: 1) a persistent PFS benefit with VTD vs TD in the overall population, as well as in subgroups of patients with high risk and low risk MM; 2) the ability of VTD, but not of TD, incorporated into double ASCT to overcome the adverse prognosis related to t(4;14); 3) the significant contribution of VTD consolidation to improved outcomes seen for patients randomized to the VTD arm; 4) the lack of more resistant relapse after exposure to VTD as induction and consolidation therapy compared to TD. A longer follow up is required to assess the OS benefit, if any, with VTD plus double ASCT.
Cavo:Bristol-Myers Squibb: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Onyx: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Millennium: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Janssen: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees; Celgene: Consultancy, Honoraria, Membership on an entity’s Board of Directors or advisory committees. Tacchetti:Janssen and Celgene: Honoraria. Zamagni:Celgene: Honoraria; Janssen-Cilag: Honoraria. Caravita:Celgene: Honoraria, Research Funding. Brioli:Celgene: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal