Abstract
Imatinib mesylate (IM) is the first line therapy against Chronic Myeloid Leukemia (CML), effectively prolonging overall survival. Because discontinuation of treatment is associated with molecular relapse, IM is required indefinitely to maintain operational cure. To evaluate the degree of response to therapy and to highlight the persistence of the disease after treatment, patients should be monitored routinely. The gold standard for diagnosing CML is the cytogenetic analysis, a direct not-sensitive method to detect Ph-positive cells. Quantitative real-time RT-PCR (qRT-PCR) provides highly sensitive detection of BCR-ABL1 transcripts, but mRNA levels are not directly related to the number of leukemic cells and cannot detect transcriptionally silent leukemic stem cells.
Here we will propose a new sensitive approach to directly detect the number of leukemic cells using a DNA-based biomarker specific for each patient. We applied targeted next-generation sequencing for the identification of genomic BCR-ABL1 fusion junctions, and we developed a sensitive new approach to detect the number of leukemic cells by a DNA Q-PCR assay based on the genomic break-point, with a formula to calculate the number of Ph+ cells.
The percentage of the leukemic cells (LC) was calculated using the following formula: %LC= (2/(2Δct+1))*100, where ΔCt is the difference between the amplification cycles of the BCR-ABL1 and BCR reactions. The number of LC was calculated by multiplying the total number of cells analyzed in each sample by the percentage of LC calculated by the ΔCt formula. We then defined a limit of quantization and a limit of sensitivity in the evaluation of minimal residual disease (MRD), as described by guidelines for the detection of MRD by genomic Q-PCR in acute lymphoblastic leukemia (ALL). We defined a “quantitative range” of detection, the portion of the standard curve in which the MRD levels can be quantified reproducibly and accurately, and we defined the “limit of sensitivity”, the lowest MRD level that still can be detected, although not in all replicates. We thus calculated the exact number of leukemic cells only when the MRD fell within the range of quantization. The detection of MRD at the limit of sensitivity was indicated as positive but not quantified.
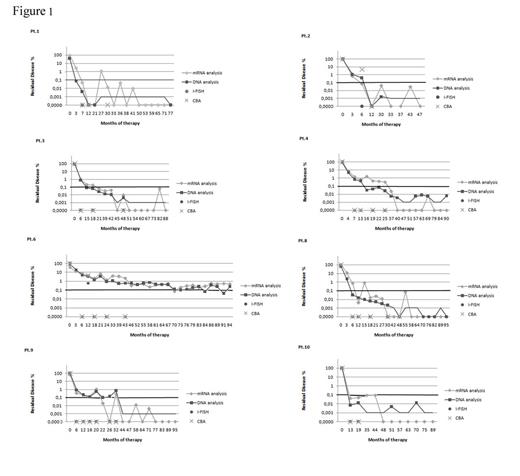
We monitored eight CML patients treated with Imatinib for 8 years. We tested the same samples by patient specific Q-PCR, and in parallel by cytogenetic analysis and by standard qRT-PCR. In all samples positive for chimeric transcripts we measured corresponding chimeric genomic DNA (gDNA) by Q-PCR, confirming the sensitivity of the Q-PCR method. According to conventional criteria, undetectable levels of BCR-ABL1 mRNA assessed by qRT-PCR are indicative of complete molecular response (CMR), but in 33.3% (45/135) samples with undetectable levels of mRNA, we detected the persistence of transcriptionally-silent leukemic cells. However, we never found samples negative by gDNA Q-PCR and positive by RNA-based qRT-PCR (Figure 1).
Thirty-six of 135 samples were also analyzed cytogenetically until the achievement of CCyR. As expected, Ph+cells were detected only in 25% (9/36) and 22,2% (8/36) of samples by CBA and I-FISH, respectively, whereas BCR-ABL1 mRNA was detected by qRT-PCR in 83.3% (30/36) of samples and Ph+ cells were detected by genomic Q-PCR in 91.7% (33/36) of samples (Figure 1).
Finally, the separation of different cell populations from blood and bone marrow revealed the presence of a population of transcriptionally silent cancer stem cells.
The gDNA based Q-PCR analysis performed on highly purified (immunomagnetically selected ) CD34+and CD3+ cells confirmed the presence of a population of transcriptionally silent cancer stem cells.
The demonstration of positive gDNA Q-PCR in 33.3% of samples negative for the RNA qRT-PCR could partially explain why some patients lose MMR and CMR and others do not, when IM is discontinued for brief periods. The gDNA based Q-PCR could be used to supplement conventional techniques, providing clinicians with additional information about disease status and response in determining whether to stop or alter therapy.
Acknowledgments to AIRC and AIL.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal