Abstract
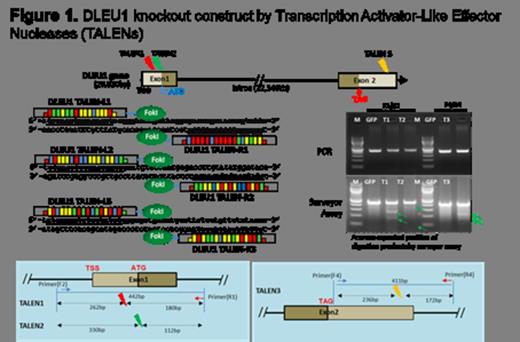
Pediatric Burkitt Lymphoma (BL) is the most common histological subtype of Non Hodgkin Lymphoma (NHL) in children and adolescents (Cairo et al, Blood, 2007; Miles/Cairo, BJHaem, 2012). We previously identified, in a subset analysis, that children with BL and a 13q deletion, particularly 13q14.3, had significantly poorer outcome and inferior overall survival (OS) despite aggressive short, intensive multi-agent chemotherapy (Poirel/Cairo et al, Leukemia, 2009; Nelson/Cairo/Sanger et al, BJHaem, 2009). Deleted in Lymphocytic Leukemia 1 (DLEU1) is a BL classifier gene on 13q14.3 region (Dave et al, NEJM, 2006). DLEU1 has been implicated in regulating programmed cell death in patients by inducing TUBB2C and UBR1 expression. Sequence-specific Transcription Activator-Like Effector Nucleases (TALENs) technologies have been developed for precision targeted genome editing in in vitro and in vivo experiments with high efficiency as a new experimental and therapeutic tool (Sander et al, Nat Bio, 2011). We have previously reported TALENs mediated transient downregulation of DLEU1 in Raji cells (Figure 1), and demonstrated a decrease in the spontaneous apoptotic rate and an increase in cell proliferation in transiently DLEU1 down-regulated Raji cells (Lee/Cairo et al, ASH, 2012).
We hypothesize that DLEU1 may act as a tumor suppressor gene and therefore investigated whether the silencing of DLEU1 results in changes in proliferation, programmed cell death and associated signaling pathways induced by Rituximab and Cyclophosphamide alone and in combination in TALEN-mediated DLEU1 knockout Raji BL cells.
A stable, single DLEU1 knockout clone (DLEU1 KO) was isolated from Raji cells transfected with DLEU1 TALENs (Lee/Cairo et al, ASH, 2012). Genomic DNA and total RNA were extracted using a DNA extraction kit (Promega) and Trizol (Invitrogen) for verification of endogenous DLEU1 gene disruption by TALENs. qRT-PCR was performed by CFX96 (Bio-rad) and the protein level was measured by western blotting. DLEU1 KO cells were plated (1x104 cells) into 48 or 96 well plates and then treated with Rituximab (0, 1, 10 and 100ug/ml) and/or Cyclophosphamide (0, 1, 5 and 10mM) and cell proliferation and apoptosis were measured by MTS (Promega) and Caspase 3/7 (Promega) assay. Statistical significance was determined by a one tailed paired Student t-test.
DLEU1 KO (mono-allelic deletion) in Raji (BL) exhibited significantly reduced DLEU1 mRNA expression compared to mock control cells (83% reduction, p<0.006). There was significantly reduced expression of c-myc, TUBB2C and UBR1 mRNA (85%, p<0.001; 60%, p<0.04; 25%, p<0.006), respectively in DLEU1 KO compared to mock control cells. DLEU1-KO showed significant reduction of Caspase 3/7 activities (25% at 24hrs, p<0.007; 33% at 48hrs, p<0.03) and significant increase in cell proliferation (20% at 24hrs, p<0.04; 25% at 48hrs, p<0.03). DLEU1-KO showed a significant increase in expression of anti-apoptotic genes, Bcl-2, Bcl-xL and Mcl-1 mRNA (14%, p<0.03; 30%, p<0.04; 17%, p<0.04), respectively and a significantly a decrease in mRNA expression of pro-apoptotic genes, Bax and Bad (18%, p<0.04; 14%, p<0.04), respectively. There was a significant increase in phosphorylation of IkBa (2.6 fold, p<0.007) and Akt (1.5 fold, p<0.05) in DLEU1 KO. There was a significant reduction in Caspase-dependent apoptosis with RTX (15% reduction, p<0.05), CTX alone (24% reduction, p<0.03), and in combination (35% reduction, p<0.03) in DLEU1-KO compared to mock control. A significant increase in cell proliferation was observed with RTX (10% increase, p<0.04), CTX alone (9.4% increase, p<0.05), and combination (7.7% increase, p<0.02) in DLEU1-KO. RTX treated DLEU1-KO showed a significant increase in expression in Bcl-2 and Bcl-xL mRNA (1.3 fold, p<0.05 and 2.0 fold, p<0.02), respectively and a significant decrease in mRNA expression of Bax (1.2 fold, p<0.03).
We demonstrate that 1) TALENs mediated DLEU1 gene knockout resulted in downregulation of DLEU1 mRNA and consequently, inhibition of BL apoptosis and increase in cell proliferation, through several mechanism(s), and 2) the silencing of DLEU1 resulted in decrease in apoptosis in RTX, CTX alone and in combination treatment. We hypothesize that deletion of DLEU1 in childhood BL may result in chemotherapy resistance secondarily to a loss of a tumor suppressor gene.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal