Abstract
The localization of memory T cells to human skin is essential for long-term immune surveillance and the maintenance of barrier integrity. Although the mechanisms controlling memory T-cell migration to peripheral tissues are poorly understood, the current paradigm includes the localized secretion of “imprinting” signals from tissue-resident dendritic cells in the draining lymph nodes. Here we show that CCR8 expression by newly activated naive T cells is regulated by skin-specific factor(s) derived primarily from epidermal keratinocytes, thereby providing a mechanism for the preferential expression of CCR8 by skin-resident memory T cells. Importantly, no such effects were observed after coculture with primary cells from skin-unrelated epithelia, including mesothelium and small intestine. The keratinocyte-derived CCR8-inducing factor(s) were soluble, and independent of vitamins A and D. Furthermore, the induction of CCR8 under these conditions correlated with an increase in cutaneous lymphocyte-associated antigen expression. Our findings challenge current tissue homing paradigms, especially those involving CCR10, and emphasize the importance of steady-state epidermis rather than tissue-resident dendritic cells in controlling the localization of memory T cells within human skin.
Introduction
The majority of lymphocytes reside in peripheral tissues, where they fulfill essential immune surveillance functions.1 Peripheral tissue immune surveillance T cells (TPS) are antigen-experienced sentinels that provide first-line defense against recurrent infections and are thought to control aberrant autoimmunity.2 Early work in animal models provided clear evidence for the presence of tissue tropism in memory T cells that correlated with the site of infection or vaccine deposition.3,4 In contrast to effector T cells, which are short-lived and can traffic to multiple unrelated sites,5 such memory T cells persist in peripheral tissues long after resolution of the primary immune response,1 exist in disequilibrium with circulating blood T cells,6,7 and provide protection against reinfection in both mice and humans.6-10 The mechanisms underlying the segregation of memory T cells to distinct, nonoverlapping sites are not well understood but appear to depend on the coordinated expression of specific adhesion molecules and chemokine receptors. Understanding this process is essential for the development of rational vaccination strategies and requires the processing of tissue samples from healthy peripheral organs.
Recent studies have implicated vitamins A and D in the control of T-cell homing to the small intestine and skin tissue.11,12 In brief, vitamin A was shown to play a crucial role in the induction of the “gut-homing” receptors CCR9 and α4β7 in murine T cells,13 and the production of active vitamin A metabolites was shown to be a feature of local CD103+ dendritic cells (DCs),14-16 findings that were subsequently replicated with human T cells.14,17 The situation for skin-homing T cells is more complicated, not least because of the fact that mouse and human skin differ profoundly in terms of histology, cellular constituents, and sun exposure. The vitamin D3 metabolite 1α,25-dihydroxyvitamin D3 was shown to induce CCR10 in human T cells, whereas murine T cells, whose CCR10 gene lacks target sites for vitamin D3 response elements, did not respond.18 Of interest, induction of CCR10 in these conditions did not correlate with induction of other skin-homing receptors, including cutaneous lymphocyte-associated antigen (CLA), and for naive T cells, was dependent on the presence of the inflammatory cytokine IL-12. Furthermore, whereas CLA expression is readily found on skin-resident T cells, CCR10 expression is largely absent.19 Although there is evidence for the involvement of CCR10 in the recruitment of effector T cells to inflamed murine skin,19,20 other studies have failed to replicate these findings.21,22 Together, these data suggest a more prominent role for CCR10 in controlling effector T-cell migration under specific inflammatory conditions as opposed to immune surveillance T-cell traffic.
We have shown previously that the majority of T cells in healthy human skin express CCR8, whereas this chemokine receptor is scarce in blood T cells and absent in intestinal tissue T cells.23 Its sole ligand, CCL1, is expressed by CD1a+ DCs, most probably Langerhans cells (LCs), present within the epidermis and, at low levels, by microvessels feeding the papillary dermis.23 These findings prompted us to suggest a role for the CCL1/CCR8 chemokine system in the control of cutaneous TPS cells.24 Human blood CCR8+ T cells are capable of producing IL-5,25 but cytokine production by their skin counterparts has not been established. Here we provide direct evidence that epidermal tissue imprints skin tropism (CCR8 and CLA expression) during the activation of naive human T cells, thereby controlling the localization of skin-selective human TPS cells.
Methods
Media and reagents
Complete RPMI (cRPMI) medium consisted of RPMI 1640 plus 2mM l-glutamine, 1% nonessential amino acids, 1% sodium pyruvate, 50 μg/mL penicillin/streptomycin, 20mM HEPES, and 10% FBS (Life Technologies). AB-RPMI consisted of cRPMI supplemented with 10% pooled human AB serum in place of FBS. Complete DMEM (cDMEM) consisted of DMEM (4.5 mg/mL glucose; Life Technologies) plus 2mM l-glutamine, 1% nonessential amino acids, 50 μg/mL penicillin/streptomycin, 20mM HEPES, and 10% FBS (Life Technologies). Defined keratinocyte serum-free medium (K-SFM), Human T-Activator CD3/CD28 Dynabeads, and AccuCheck Counting Beads were purchased from Life Technologies. All-trans retinoic acid (RA), 1α,25-dihydroxyvitamin D3, and lipopolysaccharide (S abortus equii) were purchased from Sigma-Aldrich. The pan-retinoic acid receptor antagonist LE540 was obtained from Wako Chemicals. Recombinant human IL-12 was purchased from PeproTech; rhuGM-CSF and rhuIL-4 were purchased from Miltenyi Biotec.
Blood cell isolation and culture
All research involving work with human blood and tissue samples was approved by the local Research Ethics Committee. Informed consent was obtained from each participating subject before sampling in accordance with the Declaration of Helsinki. PBMCs were isolated from healthy donors by density gradient centrifugation using Lymphoprep (Axis-Shield), according to the local ethical guidelines on experimentation with human samples. Various T-cell subsets were purified by MACS according to the manufacturer's instructions (Miltenyi Biotec). Purified T cells were isolated by negative selection using Pan T Cell Isolation Kit II. Naive T cells were further isolated by depleting CD45RO+ cells using CD45RO microbeads. Purity was evaluated by flow cytometry and ranged between 92% and 99%. Monocyte-derived DCs (MDDCs) were generated from monocytes purified by negative selection using Monocyte Isolation Kit II (Miltenyi Biotec) and cultured for 6-7 days in cRPMI supplemented with 50 ng/mL GM-CSF and 20 ng/mL IL-4. DC maturation was induced with lipopolysaccharide (100 ng/mL) for 24 hours.
Tissue cell isolation and culture
Human split skin samples (∼ 10 cm2) were excised from healthy donors undergoing mamma-reduction surgery using a dermatome (0.4-0.6 mm). Sections were cut into 1-cm2 fragments and digested in a mixture of dispase II (1.25 U/mL), collagenase D (1 mg/mL), and DNase I (20 U/mL; Roche Diagnostics) for 15-30 minutes at 37°C. The epidermis was separated from the dermis using forceps, and both the epidermis and dermis were subsequently cultured for 48-72 hours at 37°C in AB-RPMI. Single-cell suspensions of migrated cells were obtained from culture media by passing through a 40-μm pore mesh, washed, and resuspended in cRPMI. Epidermal LCs (CD1ahiCD14−), dermal CD1a+ DCs, and dermal CD14+ DCs were subsequently enriched by MACS using CD1a and CD14 microbeads (Miltenyi Biotec) or sorted using a custom-built FACSAria II (BD Biosciences) after staining with anti-CD1a–FITC and anti-CD14–PE. To obtain epidermal supernatants, freshly isolated epidermal sheets and dermal tissue (uncut) were cultured in either K-SFM or AB-RPMI for 3 days. After 3 days, supernatants were harvested, centrifuged, filtered, and added directly to T-cell cultures without prior storage.
Primary keratinocyte and fibroblast lines were generated as described previously.26 Primary keratinocytes were grown as monolayers on collagen type IV-coated flasks. Keratinocyte cultures were maintained in K-SFM (Life Technologies) and used between passage 2-4. Keratinocyte-conditioned medium was harvested from semiconfluent monolayers of primary keratinocytes after 3 days in culture. Fibroblast cultures were maintained in cDMEM.
Primary mesothelial cell cultures were prepared from specimens of human omentum obtained from patients undergoing elective abdominal surgery as described previously27 and used between passage 2-4. Primary small intestinal epithelial cells were cultured from whole small intestinal cells (DV Biologics) according to company protocols.
T-cell stimulations
Naive T cells (2.5 × 105 cells per well) were stimulated with αCD3/CD28 Dynabeads at a ratio of 1:4 (bead/cell) in cRPMI in the presence of 1α,25-dihydroxyvitamin D3 (100nM) with or without IL-12 (12.5 ng/mL), RA (200nM), LE540 (2μM), or vehicle control. In some experiments, T cells were restimulated by adding αCD3/CD28 beads with or without additives on days 3 and 6. For cocultures, T cells stimulated with αCD3/CD28 beads were added to a semi-confluent monolayer of epidermal keratinocytes or dermal fibroblasts for 6 days. Culture media for these experiments comprised a 50:50 mix of cRPMI plus K-SFM for keratinocytes or cRPMI plus cDMEM for fibroblasts. For transwell experiments, stimulated T cells were separated from keratinocyte monolayers by a 0.4-μm insert (Corning Life Sciences). To assess CCR8 expression on proliferating cells, T cells were labeled with 1μM CFSE (Life Technologies) for 4 minutes at room temperature before coculture with keratinocytes.
DC/T-cell cocultures
Purified epidermal LCs, dermal CD1a+ DCs, dermal CD14+ DCs, or mature MDDCs (1 × 104 per well) were cocultured with allogeneic naive T cells (1 × 105 per well) in round-bottom 96-well plates for 9 days in the presence or absence of keratinocyte-conditioned medium (KCM), RA (200nM), or LE540 (2μM).
Flow cytometry
Cells were acquired using a FACSCanto II (BD Biosciences) or sorted using a custom-built FACSAria II (BD Biosciences), and analyzed with FlowJo Version 9 software (TreeStar). The following monoclonal antibodies were used: αCD1a (Na1/34), αCD3 (UCHT1), αCD4 (SK3), αCD8 (SK1), αCD14 (M5E2), αCD25 (M-A251), αCD45RA (HI100), αCD45RO (UCHL1), αCD83 (HB15e), αCCR4 (1G1), αCCR7 (3D12), αβ7-integrin (FIB504), and αHLA-DR (G46-6) from BD Biosciences; αCD207 (DCGM4) from Immunotech; αCLA (HECA-452) from Miltenyi Biotec; αCCR9 (96-1), a gift from Millennium Pharmaceuticals; αCCR9 (BL/CCR9) from BioLegend; αCXCR3 (49801) and αCCR10 (314305) from R&D Systems; and αFoxP3 (236A/E7) from eBioscience. Appropriate isotype controls were included in all cases. Streptavidin-PE or streptavidin-allophycocyanin (BD Biosciences) were used together with biotinylated αCCR8 monoclonal antibody clones 433H or 414B (ATCC) to detect CCR8 expression as reported previously.25 Live cells were gated based on their light scatter properties, the exclusion of doublets on forward scatter area/height plots, and the “dumping” of dead cells using Aqua Live/Dead staining (Life Technologies).
Chemotaxis
Transwell chemotaxis assays were performed using 5-μm pore polycarbonate filter inserts (Corning Life Sciences). Cells that migrated to the lower chamber were harvested and quantified by flow cytometry using a fixed number of AccuCheck Counting Beads (Life Technologies).
Statistics
Significance testing was performed using the Mann-Whitney U test, Dunn Multiple Comparison Test, 1-way ANOVA with Tukey posttest, and linear regression analyses in GraphPad Prism Version 5.0b (GraphPad Software). A difference between groups was considered significant when P < .05.
Results
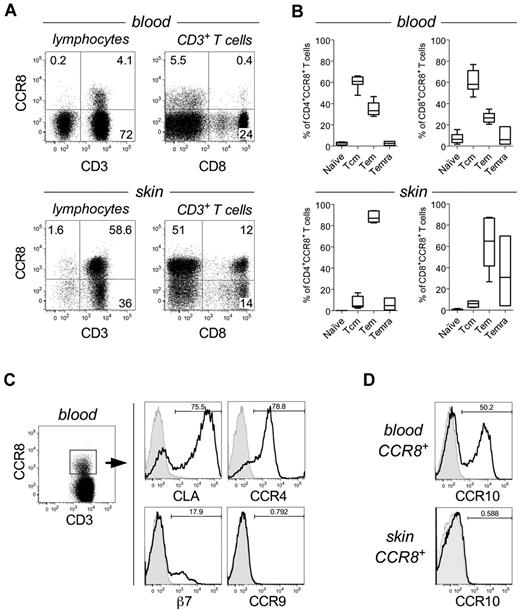
CCR8 expression in human blood leukocytes is scarce and largely restricted to central and effector memory αβ T cells25,28,29 (Figure 1A-B), despite previous reports claiming a broader distribution (reviewed by McCully and Moser24 ). In agreement with our earlier studies,23,30 we found that CCR8 is preferentially expressed on T cells present in healthy human skin (49.5% ± 5.8%; n = 7; Figure 1A) compared with those present in blood (5.05% ± 1.53%; n = 14), and have a predominant effector memory (CD45RA−CCR7−) phenotype, although for some donors a significant fraction of CD8+ T cells were terminally differentiated as characterized by CD45RA re-expression (Temra; Figure 1B). Further analysis of peripheral blood T-cell subsets revealed that, compared with CCR8− T cells, CCR8+ T cells are enriched for the skin-homing marker CLA. CCR4 was also detected at high levels, whereas expression of integrin β7 and CCR9, markers associated with gut homing, was low or absent (Figure 1C). Of interest, expression of the epidermotropic receptor CCR10 was also well represented among blood CCR8+ T cells but was not detected on skin T cells (Figure 1D). These observations suggest a unique, yet undefined, role for CCR8 in the control of cutaneous TPS cells.
CCR8 expression is enriched on skin-resident memory T cells. (A) Representative dot plots showing CCR8 expression by total lymphocytes (left) or gated CD3+ T cells (right) isolated from peripheral blood (top panels) or normal human skin tissue (bottom panels). (B) The percentage of naive (CD45RA+CCR7+), central memory (CD45RA−CCR7+; Tcm), effector memory (CD45RA−CCR7−; Tem), or RA-expressing T effector memory (CD45RA+CCR7−; Temra) within gated CD4+CCR8+ (left) or CD8+CCR8+ T cells (right) from peripheral blood (n = 7; top panels) or skin tissue (n = 6; bottom panels). Horizontal lines indicate the median. (C) Representative histograms for CLA, CCR4, integrin β7, and CCR9 expression by gated CD3+CCR8+ peripheral blood T cells. (D) Representative histograms for CCR10 expression by gated CD3+CCR8+ peripheral blood (top) or skin-resident T cells (bottom).
CCR8 expression is enriched on skin-resident memory T cells. (A) Representative dot plots showing CCR8 expression by total lymphocytes (left) or gated CD3+ T cells (right) isolated from peripheral blood (top panels) or normal human skin tissue (bottom panels). (B) The percentage of naive (CD45RA+CCR7+), central memory (CD45RA−CCR7+; Tcm), effector memory (CD45RA−CCR7−; Tem), or RA-expressing T effector memory (CD45RA+CCR7−; Temra) within gated CD4+CCR8+ (left) or CD8+CCR8+ T cells (right) from peripheral blood (n = 7; top panels) or skin tissue (n = 6; bottom panels). Horizontal lines indicate the median. (C) Representative histograms for CLA, CCR4, integrin β7, and CCR9 expression by gated CD3+CCR8+ peripheral blood T cells. (D) Representative histograms for CCR10 expression by gated CD3+CCR8+ peripheral blood (top) or skin-resident T cells (bottom).
To explain the selective anatomic distribution of CCR8, we postulated that a skin-specific mechanism might regulate the expression of this chemokine receptor and direct CCR8+ T-cell localization within healthy human skin. In support of this hypothesis, and in contrast to standard inflammatory chemokine receptors,31 CCR8 was not readily induced in purified naive blood T cells by activation alone, as assessed during in vitro stimulation with either bead-coupled or plate-bound αCD3/CD28 antibodies (Figure 2A-B; data not shown). Indeed, single-round stimulations generated < 10% CCR8+ T cells, and moderate expression was only observed after repetitive stimulations (data not shown).
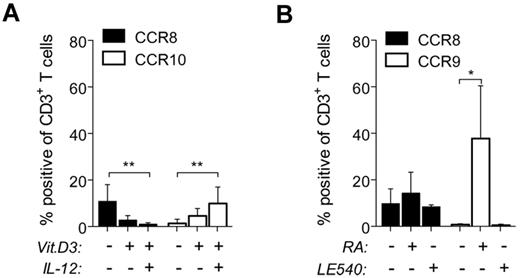
Vitamins A and D do not induce CCR8 expression. Peripheral blood naive T cells were stimulated with αCD3/CD28 beads in media alone or treated in addition with 100nM 1,25-(OH)2D3 (Vit.D3) ± IL-12 (12.5 ng/mL; A), or with 200nM RA or 2μM LE540 (B). The percentages of CD3+ T cells expressing CCR8, CCR9, or CCR10 were determined after 9 days by flow cytometry and plotted as the mean ± SD from 5-7 independent experiments. *P < .05. **P < .01.
Vitamins A and D do not induce CCR8 expression. Peripheral blood naive T cells were stimulated with αCD3/CD28 beads in media alone or treated in addition with 100nM 1,25-(OH)2D3 (Vit.D3) ± IL-12 (12.5 ng/mL; A), or with 200nM RA or 2μM LE540 (B). The percentages of CD3+ T cells expressing CCR8, CCR9, or CCR10 were determined after 9 days by flow cytometry and plotted as the mean ± SD from 5-7 independent experiments. *P < .05. **P < .01.
Recent studies have implicated vitamins A and D, and the release of their active metabolites by resident DCs, in the control of T-cell homing to the small intestine and skin tissue.11,12 We therefore examined the effect of these vitamins on CCR8 expression during in vitro stimulation of naive T cells with αCD3/CD28 beads. As expected, vitamin D3 in combination with IL-12 led to moderate CCR10 expression (Figure 2A), whereas the vitamin A metabolite RA induced CCR9 expression (Figure 2B). Of note, the minor increase in CCR8 expression induced with stimulation alone was almost completely abrogated by the addition of vitamin D3 plus IL-12 (Figure 2A), thereby revealing mutually exclusive mechanisms controlling CCR8 and CCR10 expression. Furthermore, the addition of either RA or the pan-retinoic acid receptor inhibitor LE540 did not significantly affect CCR8 expression compared with beads alone (Figure 2B). These data indicate that CCR8 expression is induced by mechanisms that are independent of vitamins A and D.
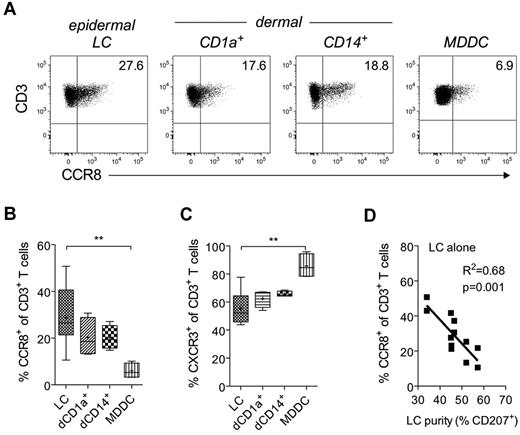
Given recent data implicating tissue-resident DCs as the main source of tissue imprinting factors,15,18 we reasoned that cutaneous DCs might be required for CCR8 expression. Indeed, epidermal LCs as well as dermal CD14+ and CD1a+ DCs all induced CCR8 expression in naive allogeneic T cells, whereas peripheral blood MDDCs were substantially less effective (Figure 3A-B). Although variable, epidermal LCs routinely turned out to be superior, in some instances inducing twice the number of CCR8+ T cells than either dermal subset (28.9% ± 12% for LCs vs 20% ± 5% for dermal CD1a+ DCs, 20% ± 2% for dermal CD14+ DCs, and 6% ± 3% for MDDCs; Figure 3B). These differences in the induction of CCR8 were not related to variations in stimulatory properties per se because CXCR3 and CCR4 were readily induced, irrespective of the type of DC used (Figure 3C; and data not shown); indeed, MDDCs induced a significantly greater percentage of CXCR3+ cells than did epidermal LCs (Figure 3C).
Induction of CCR8 expression by cutaneous DCs. Purified allogeneic naive T cells were cocultured for 9 days with MACS-enriched epidermal LCs, dermal CD1a+ DCs, dermal CD14+ DCs, or mature MDDCs at a ratio of 10:1. (A) Representative dot plots for CCR8 expression on gated CD3+ T cells. (B-C) Combined expression data for CCR8 and CXCR3 from 2-6 independent experiments. Horizontal lines indicate the median; and +, the mean. **P < .01, comparing T cells stimulated with LCs versus MDDCs. (D) The percentage of CD3+ T cells expressing CCR8 after coculture for 9 days with epidermal LCs from each donor (n = 12) is plotted against LC purity as defined by the percentage of epidermal cells expressing CD207 (Langerin) after MACS enrichment.
Induction of CCR8 expression by cutaneous DCs. Purified allogeneic naive T cells were cocultured for 9 days with MACS-enriched epidermal LCs, dermal CD1a+ DCs, dermal CD14+ DCs, or mature MDDCs at a ratio of 10:1. (A) Representative dot plots for CCR8 expression on gated CD3+ T cells. (B-C) Combined expression data for CCR8 and CXCR3 from 2-6 independent experiments. Horizontal lines indicate the median; and +, the mean. **P < .01, comparing T cells stimulated with LCs versus MDDCs. (D) The percentage of CD3+ T cells expressing CCR8 after coculture for 9 days with epidermal LCs from each donor (n = 12) is plotted against LC purity as defined by the percentage of epidermal cells expressing CD207 (Langerin) after MACS enrichment.
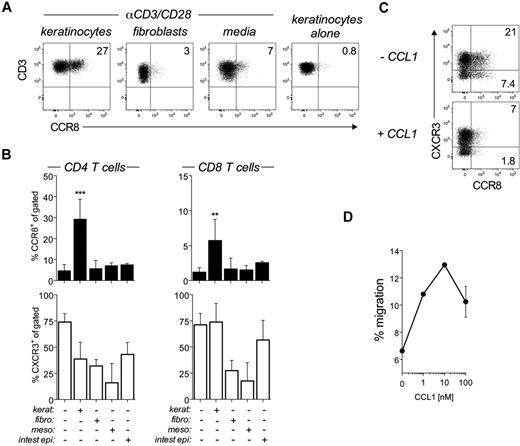
Unexpectedly, we observed that the CCR8-inducing capacity of LCs was inversely correlated with LC purity following magnetic bead isolation (MACS; Figure 3D). Consistent with this observation, LCs sorted to high purity by flow cytometry were poor inducers of CCR8 expression (< 10%) despite their potent T-cell activation properties (data not shown), which suggests that control of CCR8 expression is not a property intrinsic to LCs or any other subset of DCs. As keratinocytes were the major contaminants in MACS-isolated LC preparations (28%-42%), we hypothesized that these cells were responsible for the production of CCR8-inducing factor(s). Indeed, coculture of αCD3/CD28-bead–activated naive T cells with semiconfluent cultures of primary keratinocytes led to a significant increase in CCR8 expression (Figure 4A-B). Importantly, CCR8 expression was not observed during coculture with other tissues cells, including primary dermal fibroblasts, mesothelial cells, or intestinal epithelial cells (Figure 4A-B). In addition, coculture with keratinocytes alone (ie, in the absence of αCD3/CD28 beads) failed to induce CCR8 expression (Figure 4A), highlighting the need for TCR triggering. The keratinocyte effect was much more robust in CD4+ as opposed to CD8+ T cells (Figure 4B), and CCR8 expression was detected as early as the first division in proliferating, CFSE-labeled T cells (data not shown), which contrasts with the delayed expression of CCR9 or CCR10 in human T cells.17,18 The functionality of induced CCR8 was verified by receptor internalization on treatment with recombinant CCL1 (Figure 4C) and by dose-dependent migration to CCL1 (Figure 4D).
Regulation of CCR8 expression by epidermal keratinocytes. (A-B) Peripheral blood naive T cells were left unstimulated (keratinocytes alone) or stimulated with αCD3/CD28 beads for 6 days alone (media) or in coculture with epidermal keratinocytes (kerat), dermal fibroblasts (fibro), mesothelial cells (meso), or small intestinal epithelial cells (intest epi). (A) Representative flow cytometric dot plots showing CCR8 expression on gated CD3+ T cells after 6 days. (B) Combined expression data for CCR8 and CXCR3 by CD4+ (left panels) and CD8+ T cells (right panels) from all cocultures are plotted as mean ± SD from 2-4 independent experiments. **P < .01, comparing cocultures versus beads alone. ***P < .001, comparing cocultures versus beads alone. (C) Naive T cells cocultured with primary keratinocytes for 6 days were harvested and either left untreated or treated with 100nM CCL1 for 30 minutes at 37°C before staining for CCR8 expression. Representative dot plots of CCR8 versus CXCR3 expression on gated CD3+ T cells. (D) Transwell migration of CD4+ T cells toward 0, 1, 10, or 100nM CCL1 after activation in the presence of keratinocytes for 6 days. The numbers of migrating cells or calculated as a percentage of input cells and plotted as mean ± SD. Results shown are representative of 3 independent experiments with similar results.
Regulation of CCR8 expression by epidermal keratinocytes. (A-B) Peripheral blood naive T cells were left unstimulated (keratinocytes alone) or stimulated with αCD3/CD28 beads for 6 days alone (media) or in coculture with epidermal keratinocytes (kerat), dermal fibroblasts (fibro), mesothelial cells (meso), or small intestinal epithelial cells (intest epi). (A) Representative flow cytometric dot plots showing CCR8 expression on gated CD3+ T cells after 6 days. (B) Combined expression data for CCR8 and CXCR3 by CD4+ (left panels) and CD8+ T cells (right panels) from all cocultures are plotted as mean ± SD from 2-4 independent experiments. **P < .01, comparing cocultures versus beads alone. ***P < .001, comparing cocultures versus beads alone. (C) Naive T cells cocultured with primary keratinocytes for 6 days were harvested and either left untreated or treated with 100nM CCL1 for 30 minutes at 37°C before staining for CCR8 expression. Representative dot plots of CCR8 versus CXCR3 expression on gated CD3+ T cells. (D) Transwell migration of CD4+ T cells toward 0, 1, 10, or 100nM CCL1 after activation in the presence of keratinocytes for 6 days. The numbers of migrating cells or calculated as a percentage of input cells and plotted as mean ± SD. Results shown are representative of 3 independent experiments with similar results.
Physical separation of bead-activated T cells and keratinocytes in 2-chamber transwell culture dishes did not abrogate CCR8 expression (Figure 5A), demonstrating that soluble, as opposed to membrane-bound, factor(s) were responsible for CCR8 induction. To test this further, we cocultured allogeneic naive T cells with either MDDCs or epidermal LCs in media alone or media supplemented with 50% KCM. As expected, MDDCs on their own had no effect; however, in the presence of KCM, CCR8 was induced in up to a third of all T cells (Figure 5B). Addition of KCM to LC cultures had a minor, but significant, effect (Figure 5B). Of note, the CCR8-inducing capacity of high-purity LC preparations was “rescued” by the addition of KCM (data not shown), in full agreement with the observed effect of contaminating keratinocytes (Figure 3D). Interestingly, addition of KCM also triggered the expression of CLA, the E/P-selectin ligand required for the extravasation of blood lymphocytes to human skin tissue, while at the same time suppressing CCR4 expression (Figure 5C), regardless of whether the cells were stimulated with epidermal LCs or dermal DCs (Figure 5C). In contrast, KCM did not modulate CXCR3 expression (Figure 5C).
Keratinocyte-induced CCR8 expression is mediated by soluble factors. (A) Naive T cells were stimulated with αCD3/CD28 beads for 6 days either in direct contact with a monolayer of epidermal keratinocytes (together) or separated from the keratinocyte monolayer by a 0.4-μm transwell insert (separate). After 6 days, CCR8 expression was analyzed by flow cytometry. Data are plotted as the mean ± SEM for gated CD3+, CD4+, and CD8+ T cells from 2 independent experiments. (B-C) Purified allogeneic naive T cells were cocultured for 9 days with MACS-enriched epidermal LCs, dermal CD1a+ DCs, dermal CD14+ DCs, or MDDCs at a ratio of 10:1 in the absence or presence of KCM. (B) The percentage of T cells expressing CCR8 after 9 days in coculture with epidermal LCs or MDDCs in media alone or media supplemented with 50% KCM are plotted as mean ± SD (n = 4-10). *P < .05. (C) Percentage of CD3+ T cells expressing CCR8, CLA, CCR4, or CXCR3 after coculture with LCs, dermal CD1a+ DCs, or dermal CD14+ DC in the absence (media) or presence of KCM are plotted as mean ± SEM from 2 or 3 independent experiments. ***P < .001
Keratinocyte-induced CCR8 expression is mediated by soluble factors. (A) Naive T cells were stimulated with αCD3/CD28 beads for 6 days either in direct contact with a monolayer of epidermal keratinocytes (together) or separated from the keratinocyte monolayer by a 0.4-μm transwell insert (separate). After 6 days, CCR8 expression was analyzed by flow cytometry. Data are plotted as the mean ± SEM for gated CD3+, CD4+, and CD8+ T cells from 2 independent experiments. (B-C) Purified allogeneic naive T cells were cocultured for 9 days with MACS-enriched epidermal LCs, dermal CD1a+ DCs, dermal CD14+ DCs, or MDDCs at a ratio of 10:1 in the absence or presence of KCM. (B) The percentage of T cells expressing CCR8 after 9 days in coculture with epidermal LCs or MDDCs in media alone or media supplemented with 50% KCM are plotted as mean ± SD (n = 4-10). *P < .05. (C) Percentage of CD3+ T cells expressing CCR8, CLA, CCR4, or CXCR3 after coculture with LCs, dermal CD1a+ DCs, or dermal CD14+ DC in the absence (media) or presence of KCM are plotted as mean ± SEM from 2 or 3 independent experiments. ***P < .001
We next sought to confirm that the CCR8-inducing factor(s) were also produced by intact epidermis. In keeping with the keratinocyte data, we found that incubation of αCD3/CD28-bead–stimulated naive T cells with supernatant derived from cultures of split skin epidermis led to a significant increase in CCR8 expression for both CD4+ and CD8+ T cells (Figure 6), even at low concentrations (10%). By comparison, CXCR3 expression, which was already induced in the absence of epidermis supernatant, was upregulated to a small but significant extent on CD8+ T cells, whereas CD4+ T cells were largely unaffected (Figure 6).
Primary epidermis instructs CCR8 expression by naive T cells. Naive T cells were stimulated with αCD3/CD28 beads for 6 days alone or in the presence of increasing concentrations of epidermal supernatants. The percentages of CD4+ and CD8+ T cells expressing CCR8 (left) and CXCR3 (right) after 6 days are plotted as mean ± SEM (n = 6) from 3 independent experiments. *P < .05. **P < .01.
Primary epidermis instructs CCR8 expression by naive T cells. Naive T cells were stimulated with αCD3/CD28 beads for 6 days alone or in the presence of increasing concentrations of epidermal supernatants. The percentages of CD4+ and CD8+ T cells expressing CCR8 (left) and CXCR3 (right) after 6 days are plotted as mean ± SEM (n = 6) from 3 independent experiments. *P < .05. **P < .01.
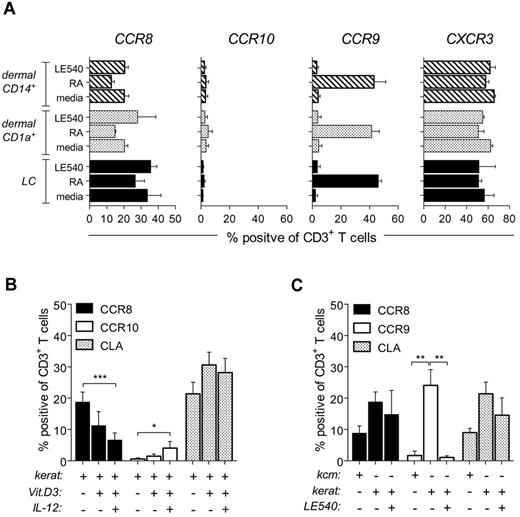
The exact nature of the CCR8-inducing factor(s) is presently unknown. Although vitamins, specifically A and D, have been implicated in the regulation of tissue homing,11,12 they do not dictate CCR8 expression. Strikingly, CCR10 expression was not detected in either allogeneic skin DC/T-cell cocultures (Figure 7A) or in cocultures containing keratinocytes plus αCD3/CD28-bead–stimulated T cells (Figure 7B). In agreement with these observations, CCR10+ cells were not detected in normal human skin (Figure 1D).19 Moreover, CCR10-inducing conditions (vitamin D3 + IL-12) actually inhibited CCR8 expression (Figure 2A), even in the presence of keratinocytes (Figure 7B). The addition of LE540, the pan-retinoic acid receptor inhibitor, to either coculture setup failed to inhibit CCR8 expression (Figure 7A-C). Unexpectedly, we did find RA-dependent CCR9 induction during keratinocyte/T-cell cocultures (Figure 7C); however, this effect was contact-dependent (data not shown) and could not be substituted using KCM (Figure 7C). Notably, RA is unstable, which emphasizes the importance of gut CD103+ DCs in the targeted delivery of RA to intestinal T cells.32
Skin-dependent regulation of CCR8 expression is independent of vitamins A and D. (A) Purified allogeneic naive T cells were cocultured for 9 days with MACS-enriched epidermal LCs, dermal CD1a+ DCs, or dermal CD14+ DCs at a ratio of 10:1 in media alone or media supplemented with 200nM RA or 2μM LE540. The percentage of T cells expressing CCR8, CCR10, CCR9, or CXCR3 was determined by flow cytometry. Data are plotted as mean ± SEM from 3 independent experiments. (B-C) Naive T cells were stimulated with αCD3/CD28 beads for 6 days in the presence of KCM (C) or a submersed monolayer of primary keratinocytes, untreated or treated with either 100nM 1,25-(OH)2D3 ± 12.5 ng/mL IL-12 (B) or 2μM LE540 (C). The percentage of total CD3+ T cells expressing CCR8, CCR9, CCR10, or CLA was determined by flow cytometry. Data are plotted as the mean ± SD (n = 4-7) from 3 independent experiments. *P < .05. **P < .01. ***P < .001.
Skin-dependent regulation of CCR8 expression is independent of vitamins A and D. (A) Purified allogeneic naive T cells were cocultured for 9 days with MACS-enriched epidermal LCs, dermal CD1a+ DCs, or dermal CD14+ DCs at a ratio of 10:1 in media alone or media supplemented with 200nM RA or 2μM LE540. The percentage of T cells expressing CCR8, CCR10, CCR9, or CXCR3 was determined by flow cytometry. Data are plotted as mean ± SEM from 3 independent experiments. (B-C) Naive T cells were stimulated with αCD3/CD28 beads for 6 days in the presence of KCM (C) or a submersed monolayer of primary keratinocytes, untreated or treated with either 100nM 1,25-(OH)2D3 ± 12.5 ng/mL IL-12 (B) or 2μM LE540 (C). The percentage of total CD3+ T cells expressing CCR8, CCR9, CCR10, or CLA was determined by flow cytometry. Data are plotted as the mean ± SD (n = 4-7) from 3 independent experiments. *P < .05. **P < .01. ***P < .001.
Consistent with the effects seen with KCM (Figure 5C), CLA was also induced on naive T cells cocultured with keratinocytes (Figure 7B-C). This finding is particularly remarkable because CLA expression did not correlate with the induction of CCR10, or CCR4, in naive human T cells either in our cultures or in previous reports,18,33 indicating that these chemokine receptors are more likely involved in effector T-cell trafficking.34
Discussion
The expression of CCR8 defines a unique subset of TPS with preferential localization within human skin tissue. Here we report that skin-resident DC subsets, including epidermal LCs and dermal DCs, failed to induce CCR8 and CLA expression in human naive T cells; however, significant expression was seen in the presence of keratinocytes or keratinocyte-conditioned medium. We further showed that these CCR8-inducing, keratinocyte-derived soluble factor(s) were released by intact epidermis and could act directly on activated T cells to induce CLA and CCR8 expression. These data contrast with the mechanism(s) underlying the imprinting of the gut-homing receptors CCR9 and α4β7, whereby the gut environment (ie, bile, stromal cells) licenses local intestinal CD103+ DCs to convert vitamin A precursors into active all-trans RA for the targeted delivery to activated lymphocytes in the draining lymph nodes.35 Although we show that human keratinocytes produced RA in sufficient quantities for induction of CCR9, RA is relatively unstable36 and thought to act primarily in an autocrine/paracrine manner.37 In agreement, CCR9 expression was expressed by T cells during coculture with keratinocytes but not when T cells were separated from keratinocytes by 2-chamber transwell membranes or when cell-free keratinocyte-conditioned medium was used. These data suggest a “short-range” activity for keratinocyte-derived RA that is unlikely to influence T-cell responses in skin-draining lymph nodes.
Interestingly, we found that the conditions favoring induction of CCR8 and CLA failed to induce CCR10 expression, a chemokine receptor associated with the preferential localization of activated T cells to skin tissue.18-20 In addition, these same conditions led to down-regulation of CCR4, a second chemokine receptor that has been associated with cutaneous T-cell traffic.20 We therefore hypothesize that CCR8 regulates the localization of TPS cells, a subset of resting memory T cells, within skin tissue in the steady state, whereas CCR10 (and CCR4) regulate the homing of activated effector T cells to skin tissue during immune responses.19,20
Our findings support a model that places healthy skin tissue at the helm of skin-tropic TPS regulation.24 It moves the attention away from local DCs, which on their own are relatively poor inducers of CCR8, and emphasizes the role of stable soluble factor(s) produced by keratinocytes that act directly on activated T cells. We propose that drainage of these factor(s) together with antigen, either in free (unprocessed) form or captured by migrating DCs, into adjacent lymph nodes results in CCR8 and CLA expression in responding T cells. Our model is supported by earlier work showing skin-dependent regulation of CLA expression.38 Alternatively, we cannot rule out a “short-range” effect in healthy skin during cutaneous T-cell contact with local DCs. It is clear, however, that at least 2 distinct processes, DC-mediated T-cell activation as well as DC-unrelated tissue factor(s), determine the imprinting of skin-tissue tropism in human TPS cells.
We hypothesize that CCR8+ TPS cells in healthy human skin represent a diverse collection of long-lived memory T cells that are unified by 2 distinct properties: (1) colocalization in a discrete peripheral tissue during steady state and (2) specificity for various antigens that have been encountered previously in the context of skin immunity. Obviously, the origin of skin antigens is wide-ranging and includes commensal as well as foreign bacteria, yeast, viruses, toxins and self-proteins, each associated with a distinct effector T-cell profile. We wish to emphasize that the localization of resting memory T cells under steady-state conditions differs from the recruitment of effector T cells to inflammatory sites. Still, it is not clear how CCR8+ TPS cells relate to effector T cells found in diverse human skin diseases. For instance, we do not know whether CCR8+ TPS cells become activated locally and turn into effector T cells or whether CCR8+ TPS cells give rise to effector T cells during engagement with antigen-presenting DCs in skin-draining lymph nodes. Preliminary data indicate that T helper cell differentiation factors (eg, IL-12) inhibit CCR8 expression, suggesting that the generation of CCR8+ TPS cells coincides with the resolution as opposed to the initiation of adaptive immune responses. As such, further studies are needed to examine the kinetics of CCR8/CLA expression in the context of the multi-stage process of T-cell activation, proliferation, and differentiation.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Ann Kift-Morgan for preparing mesothelial cell cultures.
This work was supported by the European Sixth Framework Program to the INNOCHEM consortium and The Wellcome Trust. M.L.M. is the recipient of a Wellcome Trust “Value in People” Award. B.M. is the recipient of a Royal Society Wolfson Research Merit Award.
Wellcome Trust
Authorship
Contribution: M.L.M. and B.M. conceived and designed experiments and wrote the manuscript; M.L.M. performed and analyzed experiments; K.L., S.H., and D.A.P. provided reagents/materials/analysis tools/cell sorting; and R.E.M. provided skin samples.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Bernhard Moser, Institute of Infection and Immunity, Cardiff University School of Medicine, Henry Wellcome Building, Heath Park, Cardiff CF14 4XN, United Kingdom; e-mail: moserb@cardiff.ac.uk.



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal