Abstract
Abstract 4223
SNS01-T is a novel nanoparticle that is designed to selectively initiate apoptosis in B-cell cancers such as multiple myeloma and B-cell lymphomas. SNS01-T is comprised of a plasmid encoding a pro-apoptotic form of the eukaryotic translation initiation factor 5A (eIF5A) containing a single-point mutation that prevents hypusination, an siRNA that inhibits expression of the pro-survival hypusine-eIF5A protein, and a polymer that serves to assemble the nucleic acids into a nanoparticle. SNS01-T is currently being investigated in a multi-site, open-label Phase1b/2a dose escalation study in subjects with relapsed or refractory multiple myeloma (MM).
SNS01-T and its preclinical precursors have been studied extensively in multiple myeloma and B cell lymphoma tumor models. In this study we tested the in vitro and in vivo anti-cancer activity of SNS01-T in combination with the immunomodulatory drug lenalidomide. The combination of low doses of SNS01-T and lenalidomide synergistically reduced viability of RPMI 8226 MM cells and induced apoptosis to a greater degree than either drug alone.
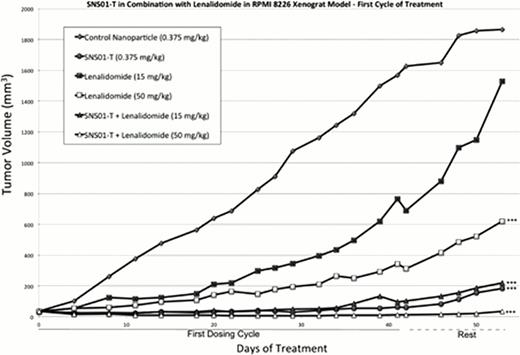
To determine whether SNS01-T treatment increases the anti-myeloma activity of lenalidomide in vivo, 0.375 mg/kg SNS01-T was combined with either 15 or 50 mg/kg lenalidomide in a RPMI 8226 xenograft model of multiple myeloma. Mice were dosed for two cycles of treatment for a total of 11 weeks of dosing. Mice with no measurable tumor at the end of the first cycle of treatment did not receive treatment in the second cycle but were monitored closely for tumor recurrence. A two-week observation period at the end of the study allowed monitoring of tumor growth after the cessation of the second cycle of treatment. At the end of the second cycle of dosing, tumor growth was inhibited by 84 % (p < 0.0001), 34 % (p = 0.05), and 98.1 % (p << 0.0001) in animals treated with SNS01-T, 50 mg/kg lenalidomide, and SNS01-T plus 50 mg/kg lenalidomide, respectively. Complete tumor regression (undetectable tumor) was achieved in 40% of mice treated with SNS01-T, 0% of mice treated with 50 mg/kg lenalidomide, and 83% of mice treated with the combination therapy of SNS01-T and 50 mg/kg lenalidomide. Complete regression of tumors treated with the combination therapy was maintained for more than 8 weeks without treatment until the end of the study in 4 of 6 (67%) of treated mice. Combining SNS01-T treatment with 50 mg/kg lenalidomide inhibited tumor growth more effectively than either drug alone and prolonged survival with 100% of mice surviving to the end of the 102-day study.
Combination therapy with SNS01-T and 15 mg/kg lenalidomide also demonstrated significant activity in a murine JMV-2 mantle cell lymphoma (MCL) xenograft model. Treatment of mice with the drug combination of SNS01-T and lenalidomide resulted in a statistically significant increase in survival compared to either SNS01-T (p = 0.002; logrank test) or lenalidomide (p = 0.007) alone.
Collectively, these preclinical studies indicate that the combination therapy of SNS01-T and lenalidomide is well tolerated, has significant activity against MM and MCL, and provides a strong rationale to evaluate SNS01-T and lenalidomide combination therapy to improve patient outcome in MM and B cell lymphomas.
Taylor:Senesco Technologies Inc.: stock options Other. Dondero:Senesco Technologies Inc.: Employment. Thompson:Senesco Technologies Inc.: Employment, Equity Ownership, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal