Abstract
Abstract 3641
Peripheral T-cell lymphoma (PTCL) is a heterogeneous group of lymphoproliferative disorders, and most subtypes have a poor prognosis even with aggressive chemotherapy. Romidepsin is a potent class 1 histone deacetylase inhibitor approved by the US Food and Drug Administration for treatment of patients with PTCL who have received at least 1 prior therapy and patients with cutaneous T-cell lymphoma who have received at least 1 prior systemic therapy. A phase 2, single-arm, open-label registration study (GPI-06–0002) demonstrated the clinical benefit and tolerability of romidepsin in patients with relapsed or refractory PTCL (data cutoff: Oct 2010). Here, we present an update of the efficacy of GPI-06–0002 and characterize patients who achieved long-term responses (≥ 12 months) as of Dec 2011 (median follow-up: 22.3 months).
Patients with histologically confirmed PTCL (N = 130) who failed or were refractory to ≥ 1 prior systemic therapy received romidepsin 14 mg/m2 as a 4-hour intravenous infusion on days 1, 8, and 15 every 28 days for up to 6 cycles; treatment could be extended for responding patients. The primary endpoint was confirmed/unconfirmed complete response (CR/CRu) determined by an independent review committee (IRC) based on the International Workshop Response Criteria. Secondary endpoints included objective response rate (ORR: CR/CRu + partial response), duration of response (DOR), and time to progression. Disease response was assessed every 2 treatment cycles. Baseline patient characteristics by DOR (≥ 12 months vs < 12 months) were examined.
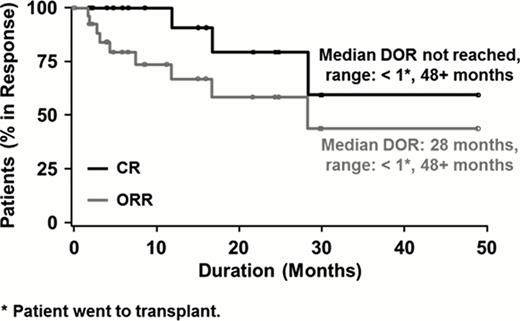
The majority of the 130 patients had stage III or IV disease (70%); 28% had bone marrow involvement. PTCL not otherwise specified (53%) and angioimmunoblastic T-cell lymphoma (21%) were the most common subtypes. Patients received a median of 2 prior systemic therapies (range, 1–8); 38% of patients were refractory to their last line of therapy. The ORR was 25% (33 of 130 patients), including CR/CRu in 15% (19 of 130) of patients. The median duration of objective response was 28 months, with the longest response ongoing at 48 months (Figure). Of the 19 patients who achieved CR/CRu, 13 (68%) had not experienced disease progression per the IRC at a median follow-up of 25.8 months. The median duration of CR had not yet been reached (range, 1–48+ months; Figure). Of the 19 patients who achieved CR/CRu, 10 were long-term responders (responses ≥ 12 months). Interestingly, heavy pretreatment (≥ 4 prior systemic therapies) did not preclude patients from achieving long-term CR/CRu: 5 of 10 patients (50%) who maintained CR/CRu for ≥ 12 months were heavily pretreated vs 1 of 9 (11%) patients with CR/CRu maintained for < 12 months. Long-term CR/CRu was achieved regardless of response to last prior therapy; only 2 of 10 (20%) long-term responders had an objective response on their last treatment. In contrast, 6 of 9 (67%) patients with CR/CRu for < 12 months responded to their last prior therapy. Furthermore, advanced disease did not preclude long-term response to romidepsin: all 10 patients (100%) who maintained CR/CRu for ≥ 12 months had stage III/IV disease vs 55.5% of those who maintained CR/CRu for < 12 months. Other characteristics, such as Eastern Cooperative Oncology Group performance status, International Prognostic Index score, age, sex, and race, were similar among patients achieving CR/CRu for ≥ 12 months or < 12 months.
Single-agent romidepsin induced durable responses in patients with relapsed/refractory PTCL, with responses ongoing at 48 months. None of the examined patient and disease characteristics predicted failure to achieve long-term remissions. These results support the use of romidepsin in relapsed/refractory PTCL.
Kaplan-Meier Plot of Duration of Objective Response and Complete Response
Coiffier:Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Pro:Allos: Honoraria; Spectrum : Honoraria; Seattle Genetics : Research Funding; Celgene: Honoraria, Research Funding. Prince:Celgene : Consultancy, Honoraria, Research Funding. Foss:Celgene : Consultancy. Sokol:Celgene : Honoraria, Speakers Bureau. Morschhauser:Celgene : Consultancy, Honoraria. Pinter-Brown:Celgene : Consultancy; Allos : Consultancy. Shustov:Celgene : Honoraria, Research Funding, Speakers Bureau. Nielsen:Celgene: Employment, Equity Ownership. Nichols:Celgene: Consultancy, Employment, Equity Ownership. Horwitz:Celgene: Consultancy, Research Funding; Kyowa Hakko Kirin Pharma: Consultancy; Bristol-Myers Squibb: Consultancy; Allos: Consultancy, Research Funding; Genzyme: Consultancy; Johnson & Johnson: Consultancy; Infinity Pharmaceuticals: Research Funding; Seattle Genetics: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal