Abstract
Abstract 2512
Tremendous progress has been made in the management of acute lymphoblastic leukemia (ALL) in children, in part, through the wide use of minimal residual disease (MRD) monitoring to guide therapeutic intensification before and after allotransplantation. Unfortunately, poor accessibility and the high costs of MRD testing have limited its use in the management of adult ALL. A universally applicable MRD quantification method has the potential to revolutionize the management of ALL in adults. Most B- and T-cell ALL patients exhibit clonal rearrangements of one or more immunoglobulin (heavy chain, IGH; light chain, IGK/IGL) or T-cell receptor (beta, B; delta, D; gamma, G) genes. Such rearrangements may be quantified in a mixture of polyclonal B or T cells by massively parallel high-throughput sequencing (HTS), enabling highly sensitive MRD quantification.
Thirty-six allografted ALL patients were selected for this retrospective study based on availability of a diagnostic sample containing leukemic cells, which was necessary for validating the amplification and sequencing method with each disease clonotype. Using Sequenta's LymphoSIGHT platform, we amplified and sequenced rearranged immunoreceptor (IR) loci from genomic DNA extracted from peripheral blood (PB) or bone marrow aspirates (BM) using V and J segment consensus primers for each IR gene (IGH, TCRB, TCRD, and TCRG) and, in some cases, D segment primers for incomplete IGH-DJ rearrangements. Sequences were analyzed using standardized algorithms for clonotype determination. Tumor-specific clonotypes were identified for each patient based on their high prevalence in a PB or BM sample at a time of high disease burden. MRD levels were then determined in serial samples of PB or BM and quantified using spiked-in reference sequences. A total of 227 samples with a median 442,672 input genomes (range 8,038 – 7,162,715) were evaluated by IR-HTS.
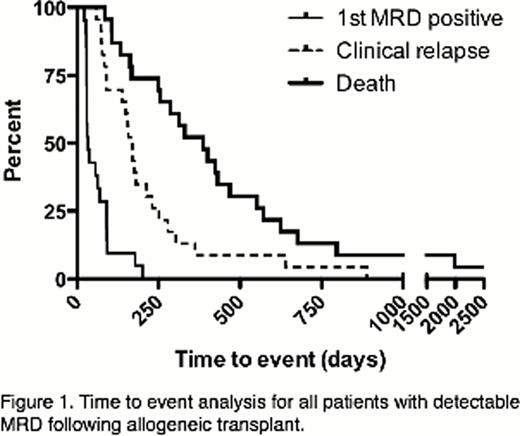
A clonal IGH sequence was identified in 17/36 (47%) ALL patients. Amongst patients who did not have a detectable IGH clonotype, 14/19 (74%) had clonal sequences at one or more other loci, including partial IGH-DJ rearrangements (5/14; 36%), TCRB (4/14; 29%), TCRD (3/14; 21%), or TCRG (6/14; 43%). In total, 31 of 36 (86%) ALL patients had a clonal IR sequence suitable for MRD quantification. In 4 of 5 cases without an identified clonal sequence, only PB, but no BM samples, were available. Twenty patients achieved MRD negativity following HCT and 11 did not. In the MRD negative group, 12/20 patients (60%) ultimately relapsed with a median time to clinical progression of 231 days (range 77–889), whereas 11/11 patients (100%) in the MRD positive group relapsed with a median time to clinical progression of 139 days (range 60–304). The 12 patients who relapsed after achieving MRD negativity showed a median time to molecular disease progression of 93 days (range 59–689 days). Of the 8 patients who maintained MRD negativity following HCT, 7 remain alive at a median 1542 days (range 1133–2557 days). All 22 patients with MRD detected following HCT relapsed and 21 (95%) died (median survival 387 days; range 85–1991 days) (Figure 1). The lead time between molecular disease detection by IR-HTS and clinical relapse was a median 69 days (range 0–207 days) with significant likelihood of MRD detection in a PB sample at least one, three, and six months (each p<0.0001) prior to clinical relapse. Amongst 50 contemporaneous PB and BM samples, 25 (50%) were concordantly MRD negative, 7 (14%) were MRD positive in BM but negative in PB, and 15 (30%) exhibited significantly higher MRD in BM versus PB (median 41-fold higher, range 1.1 – 411-fold).
We demonstrate a method for quantifying acute lymphoblastic leukemia MRD in the majority of patients using high-throughput sequencing of multiple immunoreceptor genes. IR-HTS MRD quantification provides valuable lead-time prior to clinical relapse that could provide an opportunity to apply additional therapeutic maneuvers while disease burden is low. Because MRD sensitivity in BM is significantly higher than in PB, marrow assessment may further improve the lead-time for predicting disease recurrence using IR-HTS. The dismal outcome of patients with ALL, particularly when disease burden persists after allogeneic transplant, demands further clinical study of both post-transplant MRD monitoring and novel methods for treating ALL relapse in evolution.
Faham:Sequentia Inc.: Employment. Carlton:Sequenta, Inc.: Employment. Zheng:Sequenta: Employment. Moorhead:Sequenta: Employment. Klinger:Sequenta, Inc.: Employment. Willis:Sequenta, Inc.: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal