Abstract
Abstract 2417
Primary mediastinal B-cell lymphoma (PMBL) is a subtype of diffuse large B-cell lymphoma (DLBCL) that affects predominantly young women (Swerdlow et al. 2008). Despite improvements due to addition of rituximab, which has become state of the art treatment, 20% of PMBL patients succumb to disease progression or relapse. Notably, here are currently no registered trials that are actively recruiting PMBL-patients and a better understanding of the underlying pathobiology may identify novel therapeutic targets and provide an alternative to dose escalation (Steidl and Gascoyne 2011).
BCL6 is a key germinal center B-cell transcription factor that suppresses genes involved in lymphocyte activation, differentiation, cell cycle arrest and DNA damage response gene. BCL6 is aberrantly expressed in certain DLBCL subgroups and BCL6 overexpression is sufficient for lymphomagenesis in mice (Cattoretti et al. 2005). In cellular- and murine DLBCL models, targeting of BCL6 via retroinverted BCL6 peptid inhibitor (RI-BPI) appears effective (Polo et al. 2004; Cerchietti et al. 2010). In conjunction with the relatively restricted expression pattern of BCL6, these data collectively suggest BCL6 as a candidate for targeted therapy in BCL6-positive lymphomas. Despite substantial work on BCL6 in lymphomas, the function of BCL6 in PMBL is unknown.
To address the BCL6 function in PMBL, we performed BCL6 depletion by siRNA in all three available PMBL cell lines: K1106, U-2940 and MedB-1. We found that BCL6 acts pro-proliferative and anti-apoptotic; however, PMBL models were only partially dependent on and not addicted to BCL6. Given that BCL6 expression in all PMBL cell lines is variable with a notable fraction of BCL6-negative cells, we argued that increasing the fraction of BCL6-positive cells might increase the level of BCL6-dependence. Since IL-4/STAT6 signaling upregulates BCL6 in mouse lymphocytes (Schroder et al. 2002), we treated PMBL cell lines with IL-4 (or IL-13) and, as expected, observed increased phosphorylated (p)STAT6 levels. Surprisingly, the pSTAT6 increase was not associated with higher – but with drastically lower BCL6 protein levels.
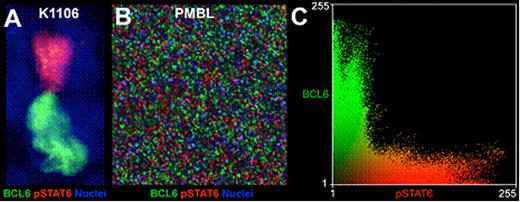
Moreover, in untreated cells, co-localization studies for pSTAT6- and BCL6 demonstrated staining in mutually exclusive subsets of cells (Figure 1A), suggesting negative interaction between BCL6 and pSTAT6. Other STAT family members were already shown to participate in the transcriptional regulation of BCL6. Thus, we examined binding of STAT6 to the proximal promoter of BCL6 in all PMBL cell lines using shift assay and chromatin immunoprecipitation. We found that STAT6 can bind all five GAS binding sites within the BCL6 promoter in vitro and in all PMBL cell lines STAT6 was bound to proximal BCL6 promoter in vivo. Furthermore, transient STAT6 depletion by siRNA and/or ectopic expression of constitutively active STAT6 confirms that pSTAT6 is sufficient for transcriptional repression of BCL6. Co-localization studies in primary patient samples demonstrated mutually exclusive BCL6/pSTAT6 distribution as a visual hallmark of the repression mechanism (Figure 1B, C).
Thus, our data demonstrate for the first time that constitutively active STAT6 transcriptionally represses BCL6 in PMBL. In conjunction with functional data, the delineated repression mechanism may prevent addiction to one single oncogenic pathway (i.e. BCL6) in PMBL.
Mutually exclusive distribution of BCL6 and pSTAT6 in PMBL
Mutually exclusive distribution of BCL6 and pSTAT6 in PMBL
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal