To the editor:
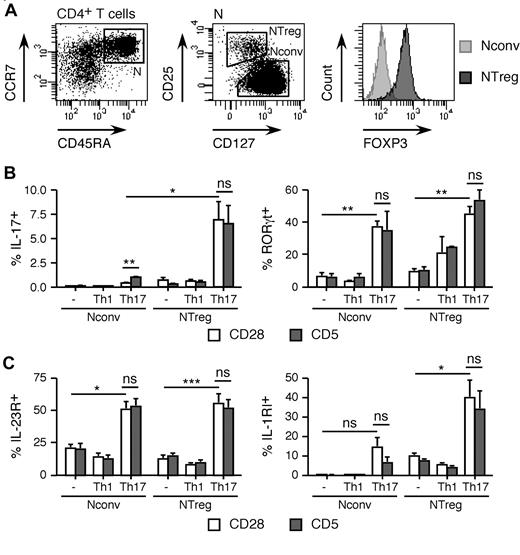
Th17 cells are a subset of CD4+ T helper cells specialized in the protection against fungi and extracellular bacteria, but also involved in the pathogenesis of autoimmune diseases. Whereas differentiation of murine Th17 is readily achieved in vitro by TCR-mediated stimulation of naive CD4+ T cells in the presence of TGF-β and IL-6, differentiation of human Th17 from naive CD4+ T cells has proved more difficult to obtain. de Wit and colleagues have recently reported in Blood that costimulation via CD5, a member of the scavenger receptor cystein-rich superfamily, rather than CD28, promotes in vitro differentiation of human Th17 from circulating naive CD4+ T cells in the presence of Th17 polarizing cytokines.1 On the other hand, we have previously reported that human Th17 are preferentially generated in vitro from NTregs, a subset of naive FOXP3+CD25+CD127− Tregs that we have previously identified,2 rather than from conventional naive FOXP3−CD25− CD4+ T cells (Nconv).3 The IL-1/IL-1RI system plays a major role in the differentiation of Th17 both in mouse models and from human NTregs.4,5 To comparatively assess the effects of costimulation via CD5 on the differentiation of Th17 from Nconv and NTreg cells, we isolated the 2 populations, ex vivo, by cell sorting (Figure 1A) and stimulated them in vitro with anti-CD3 together with anti-CD28 or anti-CD5 mAb in the presence of IL-2 alone or with Th17 or Th1 polarizing factors. As illustrated in Figure 1B, differentiation of IL-17–secreting cells from Nconv cells under Th17 polarizing conditions was moderately enhanced by costimulation via CD5, as compared with CD28, but remained overall low. Consistent with our previous data,3 differentiation of Th17 was significantly more efficiently obtained from NTregs than from Nconv cells. In addition, in contrast to Nconv cells, the proportion of Th17 differentiating from NTregs was not significantly different on costimulation via CD28 or CD5. De Wit et al proposed that costimulation via CD5 induces increased IL-23R expression, resulting in increased activation of STAT3 by IL-23 and consequent RORγt transcription. We detected significant induction of RORγt expression in both Nconv and NTreg cells stimulated under Th17 conditions as compared with neutral conditions, and to a similar extent after costimulation via CD5 or CD28 (Figure 1B). We found that IL-23R expression was increased in both Nconv and NTreg cells under Th17 polarizing conditions, but failed to detect a significant difference after costimulation via CD5 as compared with CD28 (Figure 1C). In addition, consistent with our previous findings, we detected a significantly increased expression of IL-1RI in NTregs as compared with Nconv cells, in all conditions (Figure 1C). In conclusion, efficient differentiation of NTregs to Th17 occurs after in vitro stimulation of circulating naive CD4+ T cells in the presence of Th17 polarizing factors. We observed no significant advantage to CD5 costimulation as compared with CD28. Furthermore, differentiation of human Th17 cells from conventional naive CD4+ T cells in this model system remains less efficient than that from NTregs, even in the presence of CD5 costimulation.
Human Th17 cells are preferentially generated from naive FOXP3+ Tregs on costimulation via CD28 or CD5. (A) CD4+ T cells isolated by magnetic cell sorting from healthy donors' PBMCs were stained with CD4-, CD45RA-, CCR7-, CD25-, and CD127- specific mAb and naive (N, CD45RA+CCR7+) CD4+ T cells were sorted into NTreg and Nconv populations according to CD25 and CD127 expression, as shown. Aliquots of sorted populations were stained with anti-FOXP3 mAb and analyzed by flow cytometry. (B) Sorted populations were stimulated in vitro with anti-CD2/3/28– or anti-CD2/3/5– coated beads in the absence or presence of Th1 (IL-12, 10 ng/mL) or Th17 (TGF-β, 5 ng/mL; IL-1β, 10 ng/mL; IL-23, 100 ng/mL) polarizing factors and cultured in the presence of IL-2. Aliquots of day-12 cultures were stimulated in the presence of PMA (100 ng/mL) and ionomycin (1 μg/mL) for 4 hours (the last 3 in the presence of brefeldin A, 10 μg/mL), stained with IL-17A and RORγt-specific mAb and analyzed by flow cytometry. The proportions of IL-17+ and RORγt+ cells in the cultures are shown (mean ± SEM, n = 4). (C) Aliquots of day-7 cultures, obtained as in panel B, were stained with fluorochrome-labeled anti–IL-1RI mAb or with unlabeled anti–IL-23R antibodies followed by fluorochrome-labeled goat anti–rabbit Ig antibodies and analyzed by flow cytometry. The proportions of IL-23R+ and IL-1R+ cells in the cultures are shown (mean ± SEM, n = 4).
Human Th17 cells are preferentially generated from naive FOXP3+ Tregs on costimulation via CD28 or CD5. (A) CD4+ T cells isolated by magnetic cell sorting from healthy donors' PBMCs were stained with CD4-, CD45RA-, CCR7-, CD25-, and CD127- specific mAb and naive (N, CD45RA+CCR7+) CD4+ T cells were sorted into NTreg and Nconv populations according to CD25 and CD127 expression, as shown. Aliquots of sorted populations were stained with anti-FOXP3 mAb and analyzed by flow cytometry. (B) Sorted populations were stimulated in vitro with anti-CD2/3/28– or anti-CD2/3/5– coated beads in the absence or presence of Th1 (IL-12, 10 ng/mL) or Th17 (TGF-β, 5 ng/mL; IL-1β, 10 ng/mL; IL-23, 100 ng/mL) polarizing factors and cultured in the presence of IL-2. Aliquots of day-12 cultures were stimulated in the presence of PMA (100 ng/mL) and ionomycin (1 μg/mL) for 4 hours (the last 3 in the presence of brefeldin A, 10 μg/mL), stained with IL-17A and RORγt-specific mAb and analyzed by flow cytometry. The proportions of IL-17+ and RORγt+ cells in the cultures are shown (mean ± SEM, n = 4). (C) Aliquots of day-7 cultures, obtained as in panel B, were stained with fluorochrome-labeled anti–IL-1RI mAb or with unlabeled anti–IL-23R antibodies followed by fluorochrome-labeled goat anti–rabbit Ig antibodies and analyzed by flow cytometry. The proportions of IL-23R+ and IL-1R+ cells in the cultures are shown (mean ± SEM, n = 4).
Authorship
Acknowledgments: The study was supported by the Institut National du Cancer (France) and the Cancéropôle Île-de-France (France). C.R. is supported through a fellowship from the Ligue Contre le Cancer (France).
Contribution: M.A. and D.V designed research, analyzed and interpreted data, and wrote the manuscript; and C.R. performed research and analyzed data.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Maha Ayyoub and Danila Valmori, Inserm U1102, Institut de Cancérologie de l'Ouest, 44800 Nantes-Saint Herblain, France; e-mail: maha.ayyoub@univ-nantes.fr and danila.valmori@univ-nantes.fr.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal