Abstract
Abstract 1583
Splenic Marginal Zone Lymphoma (SMZL) is a distinct lymphoma included in the WHO 2008 classification. Ann Arbor staging system is not adequate to stratify SMZL patients into risk groups, as blood and bone marrow are usually involved. Further, there are neither established criteria for starting treatment nor gold standard treatment. Herein we report an international retrospective study on 593 SMZL patients aimed to identify which factors could influence starting treatment and which could allow stratify risk categories.
Logistic regression was used to identify the factors that have conditioned treatment in the whole series and Cox regression for factors influencing Lymphoma Specific Survival (LSS) in a training set of 336 patients. The variables used were age, hemoglobin level and platelet count (as continuous variables) and sex, low albumin serum level (<3 gr/dl), high LDH and β2microglobulin, serum monoclonal component, DAT positivity, active autoimmune anemia, HCV positivity, lymphadenopathy (other than in hepatic and splenic hila) and extranodal involvement. The final model for LSS was used to produce a prognostic index (PI) and allowed to divide the patients in three risk groups applying cutpoints at the 20th and 80th centiles in patients with events. The resulting stratification was validated in another set of 227 patients. Also, the stratification was compared with the Interguppo Italiano Linfomi (IIL) score in the set of 450 patients in whom the required data were available, using the extension net reclassification improvement (NRI).
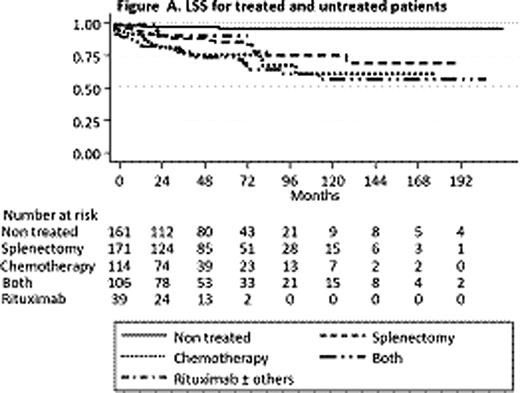
Hemoglobin (p=0.000), extrahilar lymphadenopathy (p=0.000), and HCV positivity (p=0.001), were the factors independently conditioning treatment. Untreated patients had a significant longer LSS than treated patients (p=0.000). In the treated group there were no differences in LSS according to the type of therapy (splenectomy, chemotherapy, rituximab or their combinations) (Figure A). The low number of rituximab treated patients (n=39) precluded statistically reliable conclusions. In the training set, haemoglobin (p=0.003), platelet count (p=0.043), high LDH (p=0.011) and extrahilar lymphadenopathy (p=0.020) were the factors independently influencing LSS. The resulting PI was: 0.6 × platelet count (in hundreds of thousands/mm3) + 0.2 × hemoglobin (g/dl) − 1 × high LDH (1 when LDH above normal, 0 when normal) – 0.75 × extrahilar lymphadenopathy (1 when present, 0 when absent). Applying the cut points a low, intermediate and high risk groups with significantly different 5 year LSS of 0.94, 0.78 and 0.67, respectively, were identified (Figure B). In the validation set the system also separated 3 groups with significant different 5 year LSS of 0.96, 0.88 and 0.44, respectively. This stratification was named HPLL after the determinant factors (H emoglobin, P latelet count, high L DH, L ymphadenopathy) and the resulting risk groups HPLLA, HPLLB and HPLLC. In the comparison HPLL/ABC system separated 3 groups with significantly different LSS, whereas in the IIL score there was no significant difference between the low and intermediate risk groups (p=0,102). Using as reclassification calibration statistics the Hosmer-Lemeshow test, HPLL/ABC (χ2 =1.48; p=0.475) showed a better fit than the IIL score (χ2= 3.339; p=0.118) indicating that HPLL/ABC discriminated better the risk groups.
In this large SMZL series, haemoglobin, extrahilar lymphadenopathy and HCV positivity were the factors independently conditioning treatment. The combination of hemoglobin, platelet count, high LDH and extrahilar lymphadenopathy stratify the patients in three groups with significantly different outcome. This system may be helpful to select risk-tailored treatment approaches.
Montalban:Red Temática de Investigación Cooperativa en Cancer (RETICC): Research Funding; Asociación Española contra el Cancer: Research Funding. Mollejo:Red Temática de Investigación Cooperativa en Cancer (RETICC): Research Funding; Asociación Española contra el Cancer: Research Funding. Piris:Red Temática de Investigación Cooperativa en Cancer (RETICC): Research Funding; Asociación Española contra el Cancer: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal