Abstract
Abstract 687
Available data from published studies are inconclusive regarding the curative potential of high dose chemotherapy and autologous stem cell transplantation (HDCT/ASCT) for follicular lymphoma (FL), however, these studies often include heavily pre-treated, multiply relapsed patients who may have indolent lymphoma histology other than FL, and typically do not report long term follow-up post-ASCT, particularly in the Rituximab era. Our objective was to evaluate long term outcomes and prognostic factors associated with the use of ASCT exclusively for FL patients (pts).
We conducted a retrospective analysis of the first 100 consecutive pts with relapsed or refractory follicular lymphoma treated with HDCT/ASCT from 1993–2008 in Calgary (1993-1999=20, 2000–2003=33, 2004–2008=47). Prognostic factors for event-free (EFS) and overall survival (OS) were analyzed by the Kaplan-Meier method. Funded indications for Rituximab in FL changed during the study period in Calgary as follows: 1) monotherapy for relapsed FL in 2000, 2) with induction chemotherapy (R-CVP) in 2004, and 3) maintenance therapy in 2007.
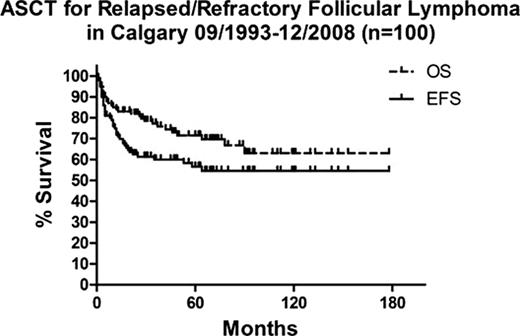
Patient characteristics at ASCT include: median age 51 years (yrs) (30-71), age >60 yrs (n=24), median time from diagnosis to ASCT 38 months (mo)(4-190), status relapsed (n=79) or refractory (n=21), CR1<1 yr (n=60), number prior failed chemotherapy regimens (1=62, 2=27, ≥3=11), prior radiotherapy (n=21), transformed disease (n=23), stage 3–4 (n=73), marrow involvement (n=31), elevated LDH (n=33), FLIPI 2–5 at ASCT (n=64), Rituximab with mobilization regimen (n=51), Rituximab with mobilization or within 6 mo prior to ASCT (n=68, including 0 pts (0%) ASCT 1993–1999, 20 pts (61%) ASCT 2000–2003, and 47 (100%) ASCT 2004–2008), conditioning with Melphalan TBI (n=40) or HDCT without TBI (n=60 [BEAM=19, Fludarabine/Busulfan=19, Melphalan=14, Zevalin® Melphalan=5, other=3]). With a median follow-up of 63 mo (16-178) post-ASCT, 38 patients have relapsed, 23 of whom died of relapsed lymphoma. Severe toxicity included 2 treatment-related deaths, and 4 deaths from secondary AML/MDS. EFS (including relapse or death from any cause) and OS at 5 yrs were 57% (95%CI 47–57) and 71% (60-80) respectively, and at 10 yrs are projected to be 55% (44-66) and 63% (49-74) respectively. EFS and OS curves start to plateau between 6–8 yrs post-ASCT as depicted in the attached figure. Each pt characteristic listed above was evaluated in univariate analysis as a potential predictor of EFS, but the only factors associated with improved EFS were: 1) ASCT 2000–2008 vs. 1993–1999 (62% vs. 40%, p=0.024, HR 0.398 [0.178-0.885]), 2) Rituximab <6 mo prior to ASCT (63% vs. 44%, p=0.012, HR 0.418 [0.211-0.828]), and 3) FLIPI score 0–1 vs. 2–5 at ASCT (65% vs. 48%, p=0.028, HR 0.499 [0.269-0.927]). FLIPI 0–1 at ASCT remained predictive of improved 5 yr EFS within the subgroup of 80 pts transplanted in 2000–2008 (75% vs. 58%, logrank p=0.051, Wilcoxon p=0.021, HR 0.461 [0.212-1.004]). There was a progressive increase in EFS following ASCT corresponding to change in Rituximab indication for FL in Calgary (5 yr EFS 40% for ASCT 1993–1999, 54% for ASCT 2000–2003, and 69% for ASCT 2004–2008 [logrank p=0.048]).
Our data support the curative potential of HDCT/ASCT when used for selected FL patients who have failed 1–2 prior chemotherapy regimens. Improvement in EFS for patients treated since 2000 are likely related to the more frequent use of Rituximab within 6 months of ASCT, either with stem cell mobilization or preceding re-induction therapy. This may be related to improved “in vivo” purging of autografts, however, this hypothesis requires further study. Finally, the FLIPI prognostic score 0–1 at ASCT predicts particularly good outcomes.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal