Abstract
Abstract 2513
The pathogenesis of ITP involves accelerated platelet destruction and decreased platelet production. The immature platelet fraction (IPF) from the Sysmex XE2100 autoanalyzer is a marker of platelet reticulocytes, so we used it to explore the mechanisms of effect of treatments of ITP. First, 67 patients with ITP were compared to 100 healthy adult controls to test the Absolute IPF (A-IPF) as a marker of thrombopoiesis. A second study investigated the plasma glycocalicin levels, a soluble external portion of membrane glycoprotein 1bα, in conjunction with A-IPF and IPF% to assess platelet turnover and survival.
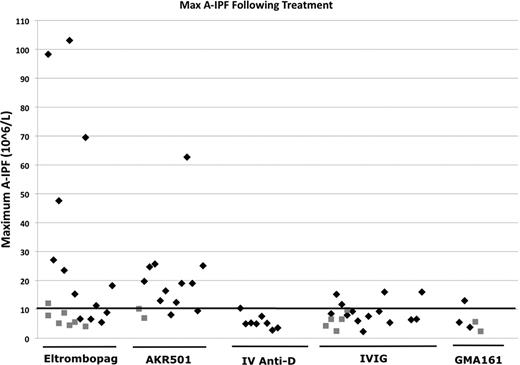
Maximum A-IPF Results for Platelet Responders (black diamonds) and Platelet Non-responders (gray squares) in ITP following therapeutic interventions.
Maximum A-IPF Results for Platelet Responders (black diamonds) and Platelet Non-responders (gray squares) in ITP following therapeutic interventions.
These results showed, as expected, that platelet responses to a TPO-A were usually associated with increased A-IPF, whereas those with an inhibitor of FcγR-mediated destruction were usually not. The effects of IV anti-D (Winrho) and IVIG in ITP were studied. 8/8 patients treated with IV anti-D had a platelet response without any A-IPF response. 10/19 patients treated with IVIG had a platelet response without a response in A-IPF, however, 4/19 had platelet and (small) A-IPF responses (figure). These results were consistent when the same patient was studied on different episodes.
In a separate study, IPF findings were compared to simultaneous samples of plasma glycocalicin levels (Beer, Blood 1994) in 21 patients. Glycocalicin Index (GCI) is a measure normalized for the platelet count. Mean plasma glycocalicin level was marginally higher in ITP patients than controls: 1.86mcg/ml (0.52–3.91) vs 1.6 mcg/ml (0.79–2.53). However, mean GCI was dramatically higher in the ITP group: 31.36 (0.368–229) vs 1.75 (0.72–2.84). For the separate treatment groups, the mean plasma glycocalicin was greatest in TPO-A treated patients and the mean GCI was greatest in IVIG & prednisolone treated patients. This illustrates the relative importance of effective platelet production and platelet turnover in these therapies. There was a statistically significant (p<0.05) inverse correlation between GCI and platelet count in ITP and there was a positive correlation between plasma glycocalicin and A-IPF, suggesting that the A-IPF was a valid marker of platelet turnover.
In summary, the results of A-IPF in ITP patients compared to healthy adult controls and in ITP patients treated with TPO-A and GMA161, demonstrate the utility of this measurement in assessing thrombopoiesis and the mechanism of treatment effect in patients with ITP. Although IVIG and IV anti-D have differing interactions with FcγR, the A-IPF results support the primary effect of anti-D and IVIG to be inhibition of platelet destruction. A small group of patients mounted both platelet and A-IPF increments to IVIG, suggesting that IVIG could also cause an increased effective platelet production. Finally, results with GCI, in a smaller number of patients, further confirmed the validity of the IPF. In the future, A-IPF could be used diagnostically (not explored in this study), for assessing response to treatment, and potentially to select the most effective treatment in individuals.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal