Abstract
Abstract 1885
Thrombocytopenia is a significant clinical problem in MDS, found in approximately 35% to 65% of pts, and is an independent risk factor for survival. Romiplostim increases platelet production by binding to and activating the thrombopoietin receptor. MDS pts who completed a romiplostim clinical study could continue romiplostim treatment in an open-label extension study. Interim results from this ongoing study are reported to provide updated safety and efficacy information on the effects of continued, long-term romiplostim treatment in pts with MDS.
MDS pts who had completed a prior romiplostim study and had platelets ≤50 × 109/L with no evidence of disease progression were eligible. Pts enrolled into the extension study from 3 prior studies with the following study designs: (1) romiplostim only for up to 52 weeks [Kantarjian JCO 2009], (2) romiplostim or placebo plus decitabine for ≥4 cycles [Greenberg ASH 2009], and (3) romiplostim or placebo plus lenalidomide for ≥4 cycles [Lyons ASH 2009]. The primary endpoint was adverse event (AE) incidence; secondary endpoints were incidence of bleeding events, platelet transfusions, and platelet response durations. On the basis of previous dosing, pts received romiplostim at 250, 500, 750, 1000, or 1500 mcg weekly, with subsequent adjustments based on platelet count. If no response was observed after 4 weeks at 1000 mcg/week, treatment was discontinued.
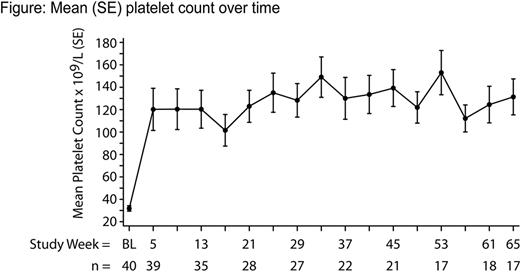
As of July 2, 2010, 40 pts had enrolled: 28 from 72 previously treated with romiplostim only, 7 from 29 previously treated with decitabine plus romiplostim or placebo, and 5 from 39 previously treated with lenalidomide plus romiplostim. In the extension study, 3 pts continued receiving decitabine and 3 pts continued receiving lenalidomide. Twenty-eight pts (70%) were male, the median age was 72 (Q1-Q3: 64–77) years, median baseline platelets were 31 × 109/L (Q1-Q3: 19–45 × 109/L), 11 pts (28%) had platelets < 20 × 109/L. At the end of their prior study, IPSS status was low (19 pts), int-1 (15), int-2 (1), or missing (5), and MDS subtypes at diagnosis were RA (11 pts), RCMD (10), MDS-U (6), RAEB-1 (8), MDS del 5q (2), RAEB-2 (1), RCMD-RS (1), and RARS (1). Twenty-one pts (53%) had bleeding events during the year before entering this study. Median duration of treatment in the extension study was 50 weeks (range: 6–134 weeks), with a median average weekly dose of 729 mcg (Q1-Q3: 593–893 mcg). The incidence of all AEs during 12-week study periods ranged from 60% to 92% and did not increase with time on study. Most AEs were mild-to-moderate; the most common being epistaxis (40%), fatigue (33%), and cough (30%). No neutralizing antibodies to romiplostim or endogenous TPO were detected. No pts died during the study; one pt died 3 months after withdrawing from the study (causality was uncertain). Peripheral blasts were increased in 2 pts (MDS-U and RA at baseline) and resolved in both cases after discontinuation of romiplostim. Three cases of progression to AML were reported, corresponding to an annual on-study event rate of 5.9% (95% CI: 1.9% to 18.3%). These pts were IPSS-risk low or int-1 at baseline and had a WHO classification of RAEB-1 or RCMD. During this study, they received 750 mcg romiplostim for 6, 49, and 36 weeks. Bone marrow blasts were 39%, 65%, and 40% at AML diagnosis. Thirty pts (75%) reported ≥1 bleeding event(s), with 17 pts (43%) reporting ≥1 clinically significant bleeding event(s) [CTCAE grade ≥3, serious AE, or any bleeding AE requiring intervention]. The proportion of pts experiencing a significant bleeding event decreased from 23% (9/40) during weeks 1–12 to 0% (0/21) during weeks 48–60. Similarly, the proportion of pts receiving a platelet transfusion decreased from 28% (11/40) during weeks 1–12 to 0% (0/21) during weeks 48–60. From week 3 onwards, the median platelet count was ≥ 50 × 109/L (Figure). Thirty-four pts (85%) had a platelet response (per IWG 2006). The median duration of platelet response was 41 weeks (Q1-Q3: 15–87 weeks).
In this study, long-term treatment of MDS pts with romiplostim demonstrated a toxicity profile consistent with published data and disease progression was consistent with expected rates. Most pts achieved a sustained platelet response while receiving romiplostim.
Fenaux:Celgene: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Janssen Cilag: Honoraria, Research Funding; ROCHE: Honoraria, Research Funding; AMGEN: Honoraria, Research Funding; GSK: Honoraria, Research Funding; Merck: Honoraria, Research Funding; Cephalon: Honoraria, Research Funding. Off Label Use: Romiplostim is approved for the treatment of thrombocytopenia in patients with chronic immune thrombocytopenia (ITP). Kantarjian:Amgen Inc.: Research Funding. Lyons:Amgen Inc.: Consultancy, Honoraria, Research Funding, Speakers Bureau. Larson:Amgen Inc.: Research Funding. Sekeres:Celgene: Consultancy, Honoraria, Speakers Bureau; Seattle Genetics: Consultancy. Becker:Amgen Inc.: Membership on an entity's Board of Directors or advisory committees, Research Funding. Muus:Celgene: Membership on an entity's Board of Directors or advisory committees; Alexion: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees. Jia:Amgen Inc.: Employment, Equity Ownership. Yang:Amgen Inc: Employment, Equity Ownership.

This icon denotes an abstract that is clinically relevant.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal