Abstract
Numerous publications have described the importance of bone morphogenetic protein (BMP) signaling in the specification of hematopoietic tissue in developing embryos. Here we investigate the full role of canonical BMP signaling in both adult and fetal liver hematopoiesis using conditional knockout strategies because conventional disruption of components of the BMP signaling pathway result in early death of the embryo. By targeting both Smad1 and Smad5, we have generated a double-knockout mouse with complete disruption of canonical BMP signaling. Interestingly, concurrent deletion of Smad1 and Smad5 results in death because of extrahematopoietic pathologic changes in the colon. However, Smad1/Smad5-deficient bone marrow cells can compete normally with wild-type cells and display unaffected self-renewal and differentiation capacity when transplanted into lethally irradiated recipients. Moreover, although BMP receptor expression is increased in fetal liver, fetal liver cells deficient in both Smad1 and Smad5 remain competent to long-term reconstitute lethally irradiated recipients in a multilineage manner. In conclusion, canonical BMP signaling is not required to maintain either adult or fetal liver hematopoiesis, despite its crucial role in the initial patterning of hematopoiesis in early embryonic development.
Introduction
During embryonic development, the hematopoietic system is one of the first complex tissues to form. Over the last 2 decades, a significant number of studies have revealed the importance of bone morphogenetic protein (BMP) signaling in mesoderm patterning and the subsequent formation of hematopoietic cells in developing embryos.1-4 In this study, we analyze the full role of canonical BMP signaling in both adult and fetal liver hematopoiesis. The necessity of BMP signaling in hematopoietic specification is largely conserved through evolution from zebrafish and Xenopus,5,6 to mouse and human.7-10 Targeted disruption of murine Bmp4, Bmp2, the type 1 receptor BmprIa, or the type 2 receptor BmprII results in early embryonic lethality and reduced formation of mesoderm.7,8,11,12 In particular, BMP4 is important in regulating hematopoiesis and has been reported to induce hematopoietic differentiation of murine and human embryonic stem cells in vitro.9,13 In addition, high concentrations of BMP4 maintain proliferation of human cord blood hematopoietic stem cells (HSCs) in vitro10 and also affect hematopoietic progenitors by inducing burst-forming units-erythroid formation of human CD34+ cells.14
BMP is a member of the transforming growth factor-β (TGF-β) superfamily. Signaling by these growth factors is initiated when binding of the ligand induces the assembly of a heteromeric complex of type 1 and type 2 serine/threonine kinase receptors. The type 1 receptors, also known as activin receptor-like kinases (ALKs), are recruited by the type 2 receptors and activate specific receptor-regulated Smads (R-Smads: Smad1-3, 5, and 8) by phosphorylation. Activated R-Smads then form complexes with the common-partner Smad4 and translocate to the nucleus where they participate in transcriptional regulation of target genes. Generally, Smad2 and 3 act downstream of the TGF-β and activin receptors, whereas Smad1, 5, and 8 primarily mediate BMP signals.15,16
In concordance with BMP ligand and receptor mutants, targeted homozygous disruption of the BMP signaling mediator Smad5 gene is also embryonic lethal. Smad5-deficient mice die between embryonic day 9.5 (E9.5) and E11.5, mainly because of defects in angiogenesis.17,18 Smad5 was shown to negatively regulate the proliferation and self-renewal of early multipotent hematopoietic progenitors derived from yolk sac and embryonic bodies in vitro.19 Although there are some controversies regarding its role in differentiation,18,19 it can be concluded that Smad5 plays a role in murine embryonic hematopoiesis, whereas the role of the related Smad1 and Smad8 is largely unknown. In zebrafish, however, loss of either Smad1 or Smad5 causes failure in generating definitive hematopoietic progenitors.20 Moreover, overexpression of Smad5 was unable to rescue the Smad1 loss-of-function phenotype. Although the role of BMP signaling in hematopoiesis has mainly been examined in embryos, a recent study demonstrated an indirect effect of BMP on the number of adult murine HSCs. By conditionally knocking out BMP receptor Ia/ALK3 in adult mice, Zhang et al showed that impaired BMP signaling increases the niche size and thereby enhances the number of HSCs.21 The same study demonstrated normal reconstitution ability of ALK3-deficient cells in wild-type (WT) recipients, although this did not address the loss of BMP signals in HSCs intrinsically, as ALK3 is not expressed by these cells.21,22
Because BMP signaling induces the HSC program during embryonic development and also influences the HSCs via their niche, it may prove useful for generating and expanding the clinically important HSCs ex vivo. As the Smads mediate signals from all of the different BMP ligands and receptors, and considering the involvement of Smad5 in hematopoietic regulation in the embryo, we have recently investigated the role of Smad5 in adult murine hematopoiesis using a Cre-mediated conditional knockout model23 with the Cre-recombinase gene under control of the interferon-inducible promoter Mx1 (MxCre).24 Surprisingly, HSCs lacking Smad5 display normal self-renewal and differentiation capacity after bone marrow (BM) transplantation, clearly indicating that Smad5 is dispensable for hematopoiesis in the adult mouse.25 Even though Smad1 and Smad5 possess inherent specificities, demonstrated by different expression and distribution patterns26-28 as well as dissimilar knockout phenotypes,17,18,27,29,30 it is conceivable that the absence of effects in our earlier study is the result of redundancy with the related Smad1. Redundant mechanisms between Smad1 and Smad5 have recently been demonstrated as Smad1+/−; Smad5+/− murine mutants, in contrast to Smad1 or Smad5 single heterozygotes, are embryonic lethal and display defects that closely resemble those seen in Smad1 or Smad5 homozygous mutants.31 In addition, Smad1 is able to rescue the Smad5−/− phenotype in zebrafish.20 Thus, Smad1 and Smad5 share equivalent functional activities in the early embryo.
To analyze the distinct versus overlapping roles of Smad1 and Smad5 in murine hematopoiesis, we created a conditional double-knockout mouse for Smad1 and Smad5. Smad8 is not expressed in adult murine HSCs,22 and is therefore not likely to play a role in regulating HSC function. Surprisingly, our results show that both Smad1 and Smad5 are dispensable for normal hematopoiesis in adult mice, as Smad1/5-deficient BM cells can compete normally with WT BM in transplantation settings and display unaffected self-renewal and differentiation capacity in vivo. In addition, fetal liver cells lacking both Smad1 and Smad5 are able to long-term reconstitute lethally irradiated recipients in a multilineage fashion, indicating that BMP signaling may be crucial only for the initiation of hematopoiesis during early embryonic development.
Methods
Mice
Mice with floxed (fl) Smad1 or Smad5 alleles for Cre-mediated knockout have been previously described (loxP sites flank exon 2 in both Smad1 and Smad5).23,32 Using the described mice, we have generated a conditional Smad1/Smad5 double-knockout on C57Bl6 background. Homozygously floxed single and double conditional knockout mice for Smad1 and Smad5 were crossed with heterozygote MxCre or VavCre mice to generate MxCre;Smad1fl/fl, MxCre;Smad1fl/fl/Smad5fl/fl, VavCre;Smad5fl/fl and VavCre;Smad1fl/fl/Smad5fl/fl offspring to study adult and fetal liver hematopoiesis, respectively. Cre expression under the influence of the Mx1 promoter was induced with 3 intraperitoneal injections (at 2-day intervals) of 250 mg of polyinositolic polycytidylic acid (pIC; Sigma-Aldrich). Cre under the influence of the Vav promoter is constitutively expressed in hematopoietic cells from approximately E10.5.33 Genotyping for detection of WT, floxed, and excised alleles was done using a 3-primer polymerase chain reaction (PCR) as described previously.23,32 The presence of Cre was verified by PCR as previously described.25 Heterozygously or homozygously floxed mice lacking Cre have 2 intact alleles of Smad1 and/or Smad5 and were hence used as WT controls. Mice were housed and bred in ventilated racks in a barrier facility. All animal experiments were approved by Lund University's Animal Ethical Committee.
Histology
Induced MxCre;Smad1fl/fl/Smad5fl/fl mice and corresponding littermate WT controls were killed when the double-knockouts started to lose weight. Organs were fixed in phosphate-buffered saline (PBS) containing 4% paraformaldehyde, embedded, and subsequently sectioned and stained with Erlish eosin for microscopic examination (Nikon Eclipse 50i; type, plan fluor; magnification ×100; numeric aperture, 0.30).
Cell preparations
Peripheral blood (PB) was collected from the tail vein and analyzed on a blood cell analyzer (Sysmex, Boule Medonice CA 530-16) to determine cell counts. Bones and spleens were crushed and passaged through a 70-μm cell strainer (BD Biosciences) to obtain single-cell suspensions. Fetal livers were dissected out at E14.5, dissociated using a syringe, and filtered through a 70-μm cell strainer. Cells were kept in PBS (Invitrogen) containing 2% fetal calf serum (Invitrogen). When necessary, red blood cells were lysed with ammonium chloride (NH4Cl; StemCell Technologies). For lineage depletion, cells were incubated with unconjugated rat antibodies against murine CD4, CD8, CD5, Gr1, Mac1, B220, and TER119 (BD Biosciences PharMingen). Lineage-positive cells were removed with a magnetic particle concentrator (MPC-6; Dynal Biotech) after incubation with sheep antirat immunoglobulin G crystallizable fragment-conjugated immunomagnetic beads (Dynal Biotech).
Flow cytometry
Rat antibodies against murine Mac1, B220, CD3, Sca-1, c-kit, CD34, FLT3, CD45.1, and CD45.2, either unconjugated or conjugated with fluorescein isothiocyanate, phycoerythrin, allophycocyanin, or PECy7 were obtained from BD Biosciences PharMingen. Unconjugated antibodies were detected with tricolor conjugated goat F(ab′)2 antirat immunoglobulin G (H+L; Invitrogen). Dead cells were excluded through staining with 7-aminoactinomycin D (Sigma-Aldrich). Cells were sorted by fluorescence-activated cell sorting (FACS) on a FACSVantage Cell Sorter (BD Biosciences) or analyzed on a FACSCalibur (BD Biosciences). Results were analyzed with FlowJo software Version 8.8.6 (TreeStar).
Hematopoietic progenitor assays
For granulocyte-macrophage colony-forming units (GM-CFU), 30 000 BM or 15 000 fetal liver cells were plated per milliliter of methylcellulose medium (M3231; StemCell Technologies) containing 50 ng/mL murine stem cell factor (Amgen), 10 ng/mL murine interleukin-3 (PeproTech), and 10 ng/mL human IL-6 (PeproTech). DNA from individual colonies was typed using PCR to check deletion efficiency.
Liquid cultures
Cells were grown in serum-free medium (StemSpan SFEM, StemCell Technologies; or X-vivo 15, BioWhittaker) supplemented with 1% bovine serum albumin (StemCell Technologies), 100 IU/mL penicillin, 100 μg/mL streptomycin (Invitrogen), 2mM l-glutamine (Invitrogen), 10−4M 2-mercaptoethanol (Sigma-Aldrich), and 50 ng/mL murine stem cell factor (Amgen), 10 ng/mL murine interleukin-3 and 10 ng/mL hIL-6 (PeproTech). Whole BM enriched for c-kit+ cells were grown at 5 × 105 cells/mL for 4 weeks. The cells were replated at 5 × 105 cells/mL and scored every third day. Lineage−, Sca-1+, c-kit+ (LSK) CD34− cells enriched for HSCs were isolated by FACS (FACSAria Cell Sorter; BD Biosciences) and plated at one cell per well in 96-well plates containing 50 μL medium. A total of 60 μL fresh supplemented medium containing cytokines was added at day 7, and proliferating cell clones were scored after 12 days (low proliferating < 200 cells, high proliferating > 200 cells).
Cell-cycle analysis
Freshly isolated BM cells were enriched for the c-kit+ population using magnetic beads (Fisher Scientific) and stained with antibodies for lineage markers, Sca-1, c-kit, and CD34 before fixation using 0.4% formaldehyde (VWR) and permeabilized with 0.2% Triton-X (Sigma-Aldrich). Permeabilized cells were subsequently incubated with anti-Ki67 antibody (BD Biosciences PharMingen) and analyzed on a FACSAria cell sorter (BD Biosciences).
Real time quantitative PCR
RNA isolation and cDNA synthesis were performed as previously described22 using sorted LSK CD34− cells from the BM, or sorted Lin− Sca-1+ AA4.1+ cells from fetal liver. Quantification and normalization of RNA levels of different Smads and receptors involved in BMP signaling were also done as previously described,22 using the TaqMan System and primers (Applied Biosystems).
Transplantations
In noncompetitive transplantations of adult mice, 1 × 106 fresh BM cells from pIC-induced MxCre;Smad1fl/fl/Smad5fl/fl or WT mice were transplanted, whereas 1 × 105 or 5 × 105 BM cells from induced MxCre;Smad1fl/fl, MxCre;Smad1fl/fl/Smad5fl/fl, and WT mice were transplanted together with 5 × 105 B6/SJL competitor BM cells in the competitive setting. When studying embryonic hematopoiesis, 2 × 106 unfractionated E14.5 fetal liver cells from VavCre;Smad5fl/fl, VavCre;Smad1fl/fl/Smad5fl/fl, or WT embryos were transplanted without support cells. In all settings, cells were transplanted into lethally irradiated (900 cGy) recipient mice (2 or 3 recipients/donor). Donor, recipient, and support cells were tracked using congenic mouse strains expressing different isoforms of the panhematopoietic marker CD45 (CD45.1 vs CD45.2). Short-term hematopoiesis was analyzed 4 to 6 weeks after transplantation. After 16 to 40 weeks (long-term), recipients were killed and half a femur-equivalent of BM was transplanted into lethally irradiated secondary recipients. PB was collected at several time points to determine donor reconstitution levels and lineage distribution by FACS analysis.
Statistical analysis
The significance of results was analyzed using the Mann-Whitney test (nonparametric); P values less than .05 were considered significant.
Results
Concurrent deletion of Smad1 and Smad5, but not Smad1 alone, results in death because of pathology in the colon
To study the effect of Smad1 and Smad1/5 deficiency during steady-state hematopoiesis, MxCre;Smad1fl/fl, MxCre;Smad1fl/flSmad5fl/fl, and WT mice were induced with pIC at 6 to 12 weeks of age, whereafter they were monitored for weight gain as an indicator of health. Smad1-deficient mice grew normally and remained healthy until the experiment was terminated at 16 weeks after induction (Figure 1A; and data not shown), as we previously reported for Smad5-deficient mice.25 When analyzing hematopoietic parameters 8 weeks after induction, Smad1-deficient mice displayed unperturbed cell numbers and lineage distribution in PB, BM, and spleen, except for a minor increase in B cells in the spleen (Figure 1B). Smad1 knockout mice also show normal numbers of LSK cells, a population enriched for hematopoietic stem and progenitor cells (Table 1).
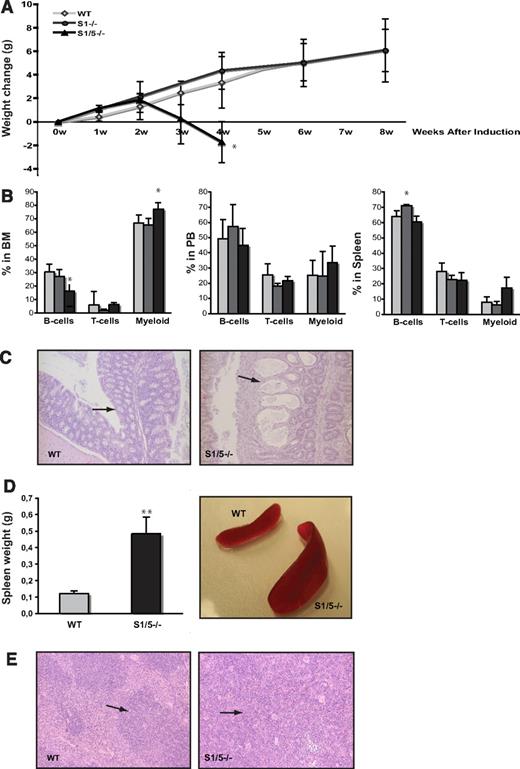
Induced deletion of Smad1 and Smad5, but not Smad1 alone, results in death because of pathology in the colon. (A) Weight change after induced deletion of Smad1 (S1−/−) and Smad1/Smad5 (S1/5−/−). (B) Lineage distribution in BM (left), PB (middle), and spleen (right) of induced mice (B cells; B220, T cells; CD3, and myeloid cells; Mac-1). Data are mean ± SD (n = 7 for WT, 3 for S1−/−, and 4 for S1/5−/−). *P < .05, **P < .001.  represent WT;
represent WT;  , S1−/−; and ▬, S1/5−/−. (C) Pathologic changes in the colon submucosa of S1/5−/− mice (right) compared with WT (left; original magnifications ×100). (D) Spleen weights are mean ± SD (n = 7) (left), representative spleens from WT and S1/5−/− mice (right). (E) Representative histologic photographs of WT (left) and enlarged S1/5−/− spleens (right; original magnifications ×100). The arrows indicate the red pulp of the spleen.
, S1−/−; and ▬, S1/5−/−. (C) Pathologic changes in the colon submucosa of S1/5−/− mice (right) compared with WT (left; original magnifications ×100). (D) Spleen weights are mean ± SD (n = 7) (left), representative spleens from WT and S1/5−/− mice (right). (E) Representative histologic photographs of WT (left) and enlarged S1/5−/− spleens (right; original magnifications ×100). The arrows indicate the red pulp of the spleen.
Induced deletion of Smad1 and Smad5, but not Smad1 alone, results in death because of pathology in the colon. (A) Weight change after induced deletion of Smad1 (S1−/−) and Smad1/Smad5 (S1/5−/−). (B) Lineage distribution in BM (left), PB (middle), and spleen (right) of induced mice (B cells; B220, T cells; CD3, and myeloid cells; Mac-1). Data are mean ± SD (n = 7 for WT, 3 for S1−/−, and 4 for S1/5−/−). *P < .05, **P < .001.  represent WT;
represent WT;  , S1−/−; and ▬, S1/5−/−. (C) Pathologic changes in the colon submucosa of S1/5−/− mice (right) compared with WT (left; original magnifications ×100). (D) Spleen weights are mean ± SD (n = 7) (left), representative spleens from WT and S1/5−/− mice (right). (E) Representative histologic photographs of WT (left) and enlarged S1/5−/− spleens (right; original magnifications ×100). The arrows indicate the red pulp of the spleen.
, S1−/−; and ▬, S1/5−/−. (C) Pathologic changes in the colon submucosa of S1/5−/− mice (right) compared with WT (left; original magnifications ×100). (D) Spleen weights are mean ± SD (n = 7) (left), representative spleens from WT and S1/5−/− mice (right). (E) Representative histologic photographs of WT (left) and enlarged S1/5−/− spleens (right; original magnifications ×100). The arrows indicate the red pulp of the spleen.
Steady-state hematopoiesis in pIC-induced mice
| Genotype . | RBC PB, ×1012/L . | WBC PB, ×109/L . | Cells/femur, ×106 . | LSK/femur, ×103 . | Cells/spleen, ×106 . | RBC spleen,† ×109 . |
|---|---|---|---|---|---|---|
| WT (n = 3-7) | 8.3 ± 0.4 | 7.5 ± 3.2 | 41.7 ± 8.3 | 132.1 ± 41.0 | 207.0 ± 80.8 | 32.5 ± 5.0 |
| Smad1 KO (n = 3) | 8.3 ± 0.2 | 10.8 ± 1.8 | 39.3 ± 2.9 | 132.7 ± 23.9 | 222.7 ± 39.6 | NA |
| Smad1/5 KO (n = 4) | 1.8 ± 0.5* | 8.1 ± 1.8 | 38.4 ± 6.9 | NA | 511.5 ± 164.3* | 77.5 ± 9.6* |
| Genotype . | RBC PB, ×1012/L . | WBC PB, ×109/L . | Cells/femur, ×106 . | LSK/femur, ×103 . | Cells/spleen, ×106 . | RBC spleen,† ×109 . |
|---|---|---|---|---|---|---|
| WT (n = 3-7) | 8.3 ± 0.4 | 7.5 ± 3.2 | 41.7 ± 8.3 | 132.1 ± 41.0 | 207.0 ± 80.8 | 32.5 ± 5.0 |
| Smad1 KO (n = 3) | 8.3 ± 0.2 | 10.8 ± 1.8 | 39.3 ± 2.9 | 132.7 ± 23.9 | 222.7 ± 39.6 | NA |
| Smad1/5 KO (n = 4) | 1.8 ± 0.5* | 8.1 ± 1.8 | 38.4 ± 6.9 | NA | 511.5 ± 164.3* | 77.5 ± 9.6* |
Data are mean ± SD.
RBC indicates red blood cell count; WBC, white blood cell count; PB, peripheral blood; and NA, not applicable.
P < .05.
Spleens suspended in 10 mL PBS.
When Smad1 and Smad5 were concurrently deleted (hereafter referred to as Smad1/5−/−), the mice began to lose weight and most mice died within 4 weeks after induction (Figure 1A). All Smad1/5−/− mice were anemic (Table 1) and tested positive for hemoglobin in the feces (data not shown), although this was not seen in any of the littermate controls. In addition to MxCre-induced gene deletion being highly efficient in the BM, the Mx1 promoter is also active in numerous other tissues, for example, the gastrointestinal tract.24 Several studies have previously shown that BMP signaling is crucial for maintenance of normal intestinal function.34,35 In addition, when we conditionally knocked out the common Smad4 in a previous study, this resulted in a dramatic colon phenotype and intestinal hemorrhage, which was hypothesized to be the result of disrupted BMP signaling.36 Accordingly, histologic examination of the gastrointestinal organs from induced WT and MxCre;Smad1fl/flSmad5fl/fl mice revealed pathologic changes in the colon submucosa of Smad1/5-deficient mice, including dilated crypts and occasional infiltration of inflammatory cells (Figure 1C). These mice also demonstrated a 4-fold increased spleen weight compared with WT controls (Figure 1D) with an accompanying increase in spleen cell numbers (Table 1), a phenotype also observed in the Smad4-deficient mice.36 The enlarged spleens displayed greatly expanded red pulp with prominent extramedullary hematopoiesis, consisting mainly of erythroid cells and megakaryocytes as determined by histologic analysis (Figure 1E). The number of red blood cells was also substantially increased in the enlarged spleens (Table 1), suggesting that the displayed splenomegaly is a response to the intestinal bleeding. These results clearly confirm that the colon pathology seen in mice lacking Smad4 is the result of disturbed BMP signaling. Moreover, FACS analysis of mature lineage-committed cells in different hematopoietic organs from induced mice revealed that Smad1/5−/− mice displayed a decrease in B cells, and an increase in myeloid cells in the BM compared with WT, which was not detected in PB or spleen (Figure 1B).
Smad1/5-deficient BM cells display decreased colony-forming ability in vitro
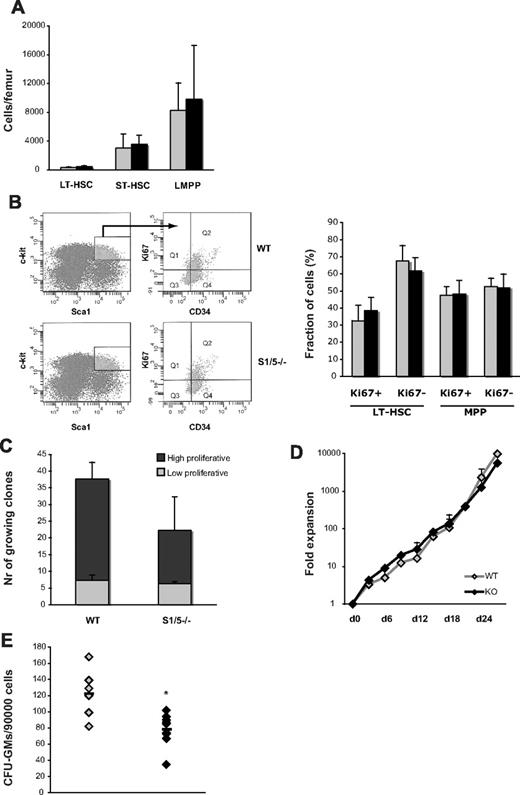
To further study the features of hematopoietic stem and progenitor cells lacking Smad1 and Smad5, BM cells were isolated from WT and VavCre;Smad1−/−Smad5−/− littermates (the latter do not develop the lethal colon phenotype seen in induced MxCre;Smad1fl/flSmad5fl/fl mice). Both Smad1 and Smad5 were 100% deleted in the HSC fraction (LSKCD34− cells) of these mice (data not shown). Importantly, the number of phenotypic long- and short-term HSCs, as well as lymphoid-primed multipotent progenitor cells remained unaltered in the absence of Smad1 and Smad5 (Figure 2A). In addition, freshly sorted long-term HSCs as well as multipotent progenitors from Smad1/5 knockout mice expressed similar levels of the proliferation marker Ki67 compared with littermate controls (Figure 2B). Furthermore, no significant difference in proliferation capacity was noticed between Smad1/5-deficient and WT cells when sorted HSCs were seeded as single cells (Figure 2C) or when the c-kit+ population, enriched for hematopoietic progenitors, from the same mice was cultured in bulk (Figure 2D). Together, these data demonstrate that Smad1/5 signaling is dispensable for HSC and progenitor number, quiescence, as well as in vitro proliferation capacity. However, when plated in methylcellulose, BM cells lacking Smad1 and Smad5 displayed a significantly decreased capacity to form myeloid colonies compared with WT (Figure 2E).
Concurrent deletion of Smad1 and Smad5 alters the colony-forming ability of myeloid progenitors in vitro. (A) Quantity of phenotypic hematopoietic stem and progenitor cells in WT and S1/5−/− mice, as determined by differential counts and FACS (LT-HSC; LSKCD34−Flt3low, ST-HSC; LSKCD34+Flt3low, lymphoid-primed multipotent progenitor; LSKCD34+Flt3high). (B) Relative fraction of dividing cells as defined by expression of Ki67 in LT-HSCs (LSKCD34−) and multipotent progenitors (LSKCD34+). Representative experiment using BM from one S1/5−/− mouse and a littermate control (left), and pooled data from 3 independent experiments (right). (C) Single-cell proliferation culture of primitive LSKCD34− cells, and (D) bulk culture of c-kit-enriched BM cells. (E) Quantity of myeloid progenitors, as determined by the number of colony-forming cells (GM-CFU). Data are mean ± SD (n = 2-5). *P < .05.  represent WT; and ▬, S1/5−/−.
represent WT; and ▬, S1/5−/−.
Concurrent deletion of Smad1 and Smad5 alters the colony-forming ability of myeloid progenitors in vitro. (A) Quantity of phenotypic hematopoietic stem and progenitor cells in WT and S1/5−/− mice, as determined by differential counts and FACS (LT-HSC; LSKCD34−Flt3low, ST-HSC; LSKCD34+Flt3low, lymphoid-primed multipotent progenitor; LSKCD34+Flt3high). (B) Relative fraction of dividing cells as defined by expression of Ki67 in LT-HSCs (LSKCD34−) and multipotent progenitors (LSKCD34+). Representative experiment using BM from one S1/5−/− mouse and a littermate control (left), and pooled data from 3 independent experiments (right). (C) Single-cell proliferation culture of primitive LSKCD34− cells, and (D) bulk culture of c-kit-enriched BM cells. (E) Quantity of myeloid progenitors, as determined by the number of colony-forming cells (GM-CFU). Data are mean ± SD (n = 2-5). *P < .05.  represent WT; and ▬, S1/5−/−.
represent WT; and ▬, S1/5−/−.
Smad1/5-deficient BM cells contribute to multilineage long-term reconstitution when transplanted into lethally irradiated recipients
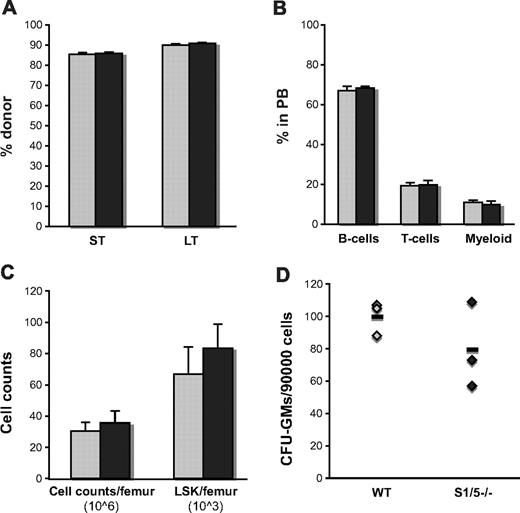
To restrict the Smad1 and Smad1/5 deletion to hematopoietic cells, BM from induced MxCre;Smad1fl/fl, MxCre;Smad1fl/flSmad5fl/fl, and WT littermate mice was transplanted into lethally irradiated recipients. Importantly, hematopoietic cells deficient for both Smad1 and Smad5 demonstrated unperturbed proliferation and differentiation potential in vivo and were able to reconstitute irradiated recipients in a multilineage fashion both short-term and long-term, as determined by FACS (Figure 3A-B). Recipients transplanted with Smad1/5−/− cells also displayed unperturbed numbers of total cells in the BM, as well as hematopoietic stem and progenitor cells per femur 5 months after transplantation (Figure 3C). When examining the numbers of myeloid progenitors in vitro, Smad1/5-deficient cells gave rise to similar numbers of colonies as WT cells, although the trend of decreased colony formation seen in the steady-state setting was still apparent in transplanted mice (Figure 3D). None of the mice transplanted with Smad1/5-deficient BM cells developed changes in the colon structure or spleen weight, indicating that the phenotype seen in the induced steady-state mice is of extrahematopoietic origin.
Smad1/5-deficient BM cells contribute to multilineage long-term reconstitution when transplanted into lethally irradiated recipients. (A) Contribution of donor cells in PB short-term (ST = 4w) and BM long-term (LT = 21w) after transplantation. (B) Distribution of myeloid cells (Mac-1), B cells (B220), and T cells (CD3) within the donor population in PB 21 weeks after transplantation presented as mean ± SD. (C) Number of total cells/femur, primitive LSK cells/femur, and (D) myeloid progenitors in BM 21 weeks after transplantation, as determined by differential counts, FACS, and the number of formed colonies (GM-CFU), respectively (n = 3 donors and 9 recipients).  represent WT; and ▬, S1/5−/−.
represent WT; and ▬, S1/5−/−.
Smad1/5-deficient BM cells contribute to multilineage long-term reconstitution when transplanted into lethally irradiated recipients. (A) Contribution of donor cells in PB short-term (ST = 4w) and BM long-term (LT = 21w) after transplantation. (B) Distribution of myeloid cells (Mac-1), B cells (B220), and T cells (CD3) within the donor population in PB 21 weeks after transplantation presented as mean ± SD. (C) Number of total cells/femur, primitive LSK cells/femur, and (D) myeloid progenitors in BM 21 weeks after transplantation, as determined by differential counts, FACS, and the number of formed colonies (GM-CFU), respectively (n = 3 donors and 9 recipients).  represent WT; and ▬, S1/5−/−.
represent WT; and ▬, S1/5−/−.
Both Smad1 and Smad1/5-deficient HSCs compete normally with WT BM cells and display unaffected self-renewal and differentiation capacity
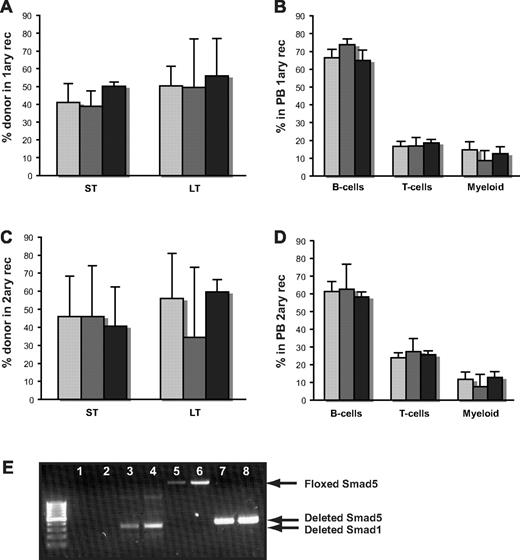
To increase the pressure on the hematopoietic system, we mixed BM from WT, Smad1−/−, and Smad1/5−/− mice with congenic WT BM at a 1:1 ratio and transplanted the cell mixture into lethally irradiated recipients. As demonstrated in Figure 4, both Smad1- and Smad1/5-deficient BM cells were able to compete normally with WT cells, and contributed to long-term multilineage reconstitution of the recipients (Figure 4A-B). Furthermore, transplantation of BM from primary to secondary recipients revealed that HSCs from both Smad1- and Smad1/5-deficient mice could reconstitute the hematopoietic system after serial transplantation and thus composed normal self-renewal capacity, as well as differentiation ability (Figure 4C-D). In addition, there was no significant difference in the number of primitive LSK cells in secondary recipients as assessed by FACS long-term after transplantation (data not shown). To avoid the risk of saturating the system by transplanting too many cells, we also performed a competitive transplantation where a reduced number of donor cells were competed against WT BM cells at a 1:5 ratio. However, in concordance with the findings from the other transplantations, the experiment revealed no difference between WT and Smad1/5−/− hematopoietic repopulation (data not shown).
Both Smad1- and Smad1/5-deficient BM cells display normal self-renewal and differentiation capacity after competitive BM transplantation. (A) Short-term contribution (ST) in PB and long-term (LT) reconstitution in BM in primary and (C) secondary recipients after competitive transplantation, as measured by Ly5.2/Ly5.1 contribution. (B) Distribution of myeloid cells (Mac-1), B cells (B220), and T cells (CD3) within the donor population in PB from primary and (D) secondary recipients analyzed by FACS long-term after transplantation. Data are mean ± SD (n = 9/5/8 donors and n = 25/15/19 recipients for WT, S1−/−, and S1/5−/−, respectively, for primary transplantations, and n = 7/5/3 donors and n = 21/15/9 recipients for WT, S1−/−, and S1/5−/−, respectively, for secondary transplantations).  represent WT;
represent WT;  , S1−/−; and ▬, S1/5−/−. (E) Representative PCR screen of individual hematopoietic colonies from WT and S1/5-deficient mice to determine deletion (total number of colonies screened, n > 240). Lanes 1 to 4 indicate floxed (no band) and deleted (300 bp) Smad1 in WT (lanes 1 and 2) and S1/5−/− colonies (lanes 3 and 4); lanes 5 to 8, floxed (2.3 kb) and deleted (387 bp) Smad5 in WT (lanes 5 and 6) and S1/5−/− colonies (lanes 7 and 8).
, S1−/−; and ▬, S1/5−/−. (E) Representative PCR screen of individual hematopoietic colonies from WT and S1/5-deficient mice to determine deletion (total number of colonies screened, n > 240). Lanes 1 to 4 indicate floxed (no band) and deleted (300 bp) Smad1 in WT (lanes 1 and 2) and S1/5−/− colonies (lanes 3 and 4); lanes 5 to 8, floxed (2.3 kb) and deleted (387 bp) Smad5 in WT (lanes 5 and 6) and S1/5−/− colonies (lanes 7 and 8).
Both Smad1- and Smad1/5-deficient BM cells display normal self-renewal and differentiation capacity after competitive BM transplantation. (A) Short-term contribution (ST) in PB and long-term (LT) reconstitution in BM in primary and (C) secondary recipients after competitive transplantation, as measured by Ly5.2/Ly5.1 contribution. (B) Distribution of myeloid cells (Mac-1), B cells (B220), and T cells (CD3) within the donor population in PB from primary and (D) secondary recipients analyzed by FACS long-term after transplantation. Data are mean ± SD (n = 9/5/8 donors and n = 25/15/19 recipients for WT, S1−/−, and S1/5−/−, respectively, for primary transplantations, and n = 7/5/3 donors and n = 21/15/9 recipients for WT, S1−/−, and S1/5−/−, respectively, for secondary transplantations).  represent WT;
represent WT;  , S1−/−; and ▬, S1/5−/−. (E) Representative PCR screen of individual hematopoietic colonies from WT and S1/5-deficient mice to determine deletion (total number of colonies screened, n > 240). Lanes 1 to 4 indicate floxed (no band) and deleted (300 bp) Smad1 in WT (lanes 1 and 2) and S1/5−/− colonies (lanes 3 and 4); lanes 5 to 8, floxed (2.3 kb) and deleted (387 bp) Smad5 in WT (lanes 5 and 6) and S1/5−/− colonies (lanes 7 and 8).
, S1−/−; and ▬, S1/5−/−. (E) Representative PCR screen of individual hematopoietic colonies from WT and S1/5-deficient mice to determine deletion (total number of colonies screened, n > 240). Lanes 1 to 4 indicate floxed (no band) and deleted (300 bp) Smad1 in WT (lanes 1 and 2) and S1/5−/− colonies (lanes 3 and 4); lanes 5 to 8, floxed (2.3 kb) and deleted (387 bp) Smad5 in WT (lanes 5 and 6) and S1/5−/− colonies (lanes 7 and 8).
To confirm efficient deletion of the Smad1 and Smad5 gene, respectively, individual hematopoietic colonies from WT, Smad1-, and Smad1/5-deficient mice were screened using PCR analysis (n > 240 colonies, Figure 4E). Consistent with previous reports testing MxCre-mediated deletion of single genes in hematopoietic colonies,25,37 the targeted exon was deleted in 100% of the colonies tested from Smad1 single knockout mice. However, when concurrently knocking out Smad1 and Smad5, the deletion efficiency was on rare occasions slightly lower of one or both of the genes, although the absolute majority of the hematopoietic cells analyzed had a complete deletion in both genes. To control that these rare hematopoietic cells with intact Smad1 or Smad5 expression did not take over and mask a potential phenotype, we sorted out donor-derived hematopoietic BM cells 16 weeks after transplantation, plated them in methylcellulose, and screened individual colonies for Smad1 and Smad5 deletion. Virtually all donor-derived cells remained deficient in both Smad1 and Smad5, verifying that the few cells with remaining Smad1 or Smad5 did not out compete the Smad1/5-deficient cells.
BMP signals can be transduced by both Smad1, 5, and 8. Although Smad8 is not expressed in primitive hematopoietic cells in the BM,10,22 we wanted to examine whether the expression levels of Smad8 were altered when knocking out Smad1 and Smad5, thus potentially generating a rescue mechanism. However, quantitative reverse-transcription (RT)–PCR analysis on sorted LSK cells from induced MxCre;Smad1fl/flSmad5fl/fl and WT mice demonstrated that Smad8 remained undetectable also in primitive Smad1/5−/− BM cells (data not shown). Hence, the lack of phenotype in Smad1/5-deficient hematopoiesis in adult mice is not because of redundancy between related intracellular Smads.
BMP signaling is dispensable for functional fetal liver hematopoiesis
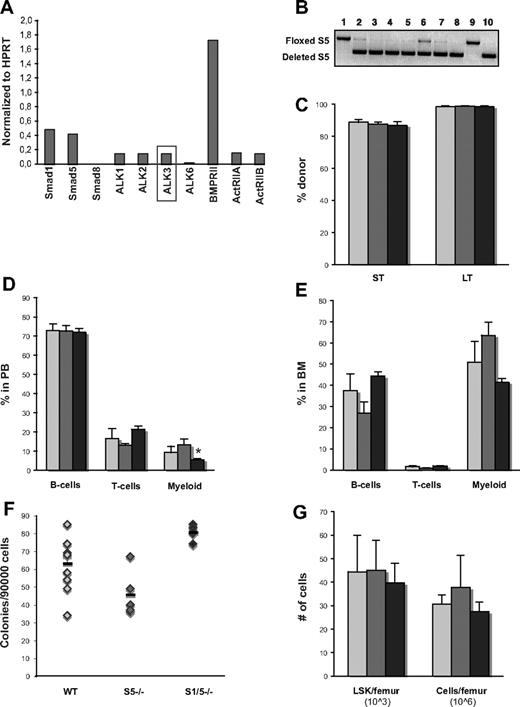
Numerous publications have described the importance of BMP signaling in the formation of hematopoietic tissue in developing embryos. Because targeted disruption of the different components of the BMP signaling pathway all result in early death of the embryo,7,8,11,12,17 the majority of these studies are based on in vitro experiments using very primitive hematopoietic cells derived from murine or human embryonic stem cells,9,13 murine yolk sac, or embryonic bodies.18,19 We therefore decided to study the role of BMP signaling in fetal liver hematopoiesis, a developmental phase in between the adult hematopoiesis in the BM and the primitive hematopoiesis found in yolk sac. To address the question of possible biologic differences between these 2 ontogenic stages, the expression level of receptors involved in BMP signaling was analyzed by quantitative RT-PCR in sorted primitive fetal liver cells. Intriguingly, the expression of ALK3 was markedly increased compared with levels in BM HSCs (Figure 5A), suggesting that there may be a discrepancy in response to BMP signaling between BM and fetal liver hematopoietic cells. As in the BM, Smad8 was not detectable in hematopoietic stem and progenitor cells derived from fetal liver.
Both Smad1 and Smad5 are dispensable for functional fetal liver hematopoiesis. (A) Expression of BMP receptors in WT fetal liver cells analyzed by quantitative RT-PCR. ALK3 is expressed in Lin− Sca-1+ AA4.1+ fetal liver but is undetectable in LSK CD34− BM cells.10,22 (B) Representative gel showing screening of Smad5 deletion (PCR) in fetal livers. Only livers with highly deleted Smad1 (data not shown) and Smad5 were used (such as #10). WT littermates lacking Cre have floxed, but undeleted, genes (such as #1). (C) Short-term (ST) donor reconstitution in PB and long-term (LT) contribution in BM. Distribution of myeloid cells (Mac-1), B cells (B220), and T cells (CD3) within the donor population in PB (D) and BM (E) analyzed by FACS long-term after transplantation. Number of myeloid progenitors (F), total cells/femur and primitive LSK cells/femur (G) in BM long-term after transplantation as determined by the number of formed colonies (GM-CFU), differential counts, and FACS, respectively. Data are mean ± SD (n = 5 donors and 15 recipients per genotype).  represent WT;
represent WT;  , S5−/−; and ▬, S1/5−/−.
, S5−/−; and ▬, S1/5−/−.
Both Smad1 and Smad5 are dispensable for functional fetal liver hematopoiesis. (A) Expression of BMP receptors in WT fetal liver cells analyzed by quantitative RT-PCR. ALK3 is expressed in Lin− Sca-1+ AA4.1+ fetal liver but is undetectable in LSK CD34− BM cells.10,22 (B) Representative gel showing screening of Smad5 deletion (PCR) in fetal livers. Only livers with highly deleted Smad1 (data not shown) and Smad5 were used (such as #10). WT littermates lacking Cre have floxed, but undeleted, genes (such as #1). (C) Short-term (ST) donor reconstitution in PB and long-term (LT) contribution in BM. Distribution of myeloid cells (Mac-1), B cells (B220), and T cells (CD3) within the donor population in PB (D) and BM (E) analyzed by FACS long-term after transplantation. Number of myeloid progenitors (F), total cells/femur and primitive LSK cells/femur (G) in BM long-term after transplantation as determined by the number of formed colonies (GM-CFU), differential counts, and FACS, respectively. Data are mean ± SD (n = 5 donors and 15 recipients per genotype).  represent WT;
represent WT;  , S5−/−; and ▬, S1/5−/−.
, S5−/−; and ▬, S1/5−/−.
Because deletion of Smad5 results in embryonic lethality at E10.5 because of defective angiogenesis, MxCre cannot be used to study fetal liver hematopoiesis as this leads to deletion also in endothelial cells. Therefore, to study the deletion of the BMP mediating Smads in fetal liver cells, we generated VavCre;Smad5fl/fl and VavCre;Smad1fl/fl/Smad5fl/fl mice, where Vav is a pan-hematopoietic gene. Driving Cre expression from the Vav promoter has been reported to cause deletion in essentially all hematopoietic cells, including HSCs with an efficiency of 93% to 96% in fetal liver while leaving endothelial cells unaffected.33 Both VavCre;Smad5−/− and VavCre;Smad1−/−/Smad5−/− mice were born at Mendelian ratios and were fertile. Fetal livers were dissected out on E14.5 and screened for deletion of respective Smad (Figure 5B). Surprisingly, when transplanted without support into lethally irradiated recipients, both Smad5- and Smad1/5-deficient fetal liver cells were capable of reconstituting the hosts both short-term and long-term (Figure 5C). They were also able to generate all mature blood lineages at levels comparable with WT cells, apart from a slight but significant reduction of myeloid cells in PB from mice receiving Smad1/5−/− cells (Figure 5D). This decrease in myeloid cells was, however, not seen in the BM, either when analyzing lineage distribution by FACS (Figure 5E) or when examining the number of myeloid colony-forming progenitors (Figure 5F). In addition, mice receiving either Smad5−/− or Smad1/5−/− BM cells exhibited normal counts of red and white blood cells (data not shown) and displayed normal numbers of total BM cells as well as primitive LSK cells per femur long-term after transplantation (Figure 5G). BM cells from primary recipients were plated in methylcellulose 18 weeks after transplantation, and screening of individual colonies revealed a persistent 100% deletion of both Smad1 and Smad5. All hematopoietic parameters measured remained normal after transplantation of Smad5−/− and Smad1/5−/− cells into secondary recipients, demonstrating that these cells possess unaffected self-renewal capacity (data not shown). We therefore conclude that BMP-signaling mediated by Smad1 and Smad5 is dispensable for functional fetal liver hematopoiesis.
Discussion
A significant body of evidence states an important role for BMP in initiating hematopoietic development in the early embryo.1-4 Because targeted disruption of components of the BMP signaling pathway results in embryonic lethality, the role of BMP signaling in adult hematopoiesis is largely unknown. Still, judging from its role in the embryo, expectations have been expressed that BMP signaling may prove useful for generating and expanding clinically important HSCs ex vivo. In accordance with other components of BMP signaling, Smad5 affects hematopoiesis in early embryonic development in vitro.18,19 However, using a conditional knockout strategy, we recently demonstrated that Smad5 is dispensable for normal hematopoiesis in the adult mouse.25 To further address the issue of possible redundancy between related BMP-mediating Smads and also potential biologic differences between ontogenic stages, we created a double conditional Smad1/Smad5 knockout mouse coupled to different Cre expression patterns, thereby enabling investigation of the full role of canonical BMP signaling in both adult and fetal liver hematopoiesis.
Our results demonstrate that concurrent deletion of Smad1 and Smad5 results in death because of intestinal hemorrhage and pathologic changes in the colon. This is in agreement with previous publications describing an important role for BMP in maintaining colon homeostasis.34-36 Moreover, Smad1/5−/− mice have a compensatory enlargement of the spleen and display a decrease in B cells and increase in myeloid cells in the BM. The latter findings indicate that simultaneous deletion of Smad1 and Smad5 may affect the differentiation capacity of hematopoietic progenitors. However, the disturbed lineage distribution in the Smad1/5−/− mice at steady state might also be explained by the finding that these mice are moribund and are in a bad general condition. Accordingly, none of the mice transplanted with Smad1/5−/− BM displayed any of the described symptoms, supporting that the observed phenotype was caused by the diseased Smad1/5-deficient environment. These results confirm our previous findings, where conditionally knocking out Smad4 resulted in a very similar colon phenotype, which was then hypothesized to be the result of disrupted BMP signaling.36 Interestingly, because knocking out only Smad1 or Smad5 did not result in disruption of the colon architecture, these related Smads seem to play a redundant role in maintaining colon homeostasis.
As opposed to a previous study where Smad5 was implied to negatively regulate the proliferation of multipotent progenitors derived from yolk sac and embryonic bodies in vitro,19 Smad1/5-deficient HSCs derived from adult BM displayed normal numbers with retained proliferation status and expansion capacity in vitro, as well as in vivo. In accordance, transplanted BM cells lacking Smad1/5 displayed unaffected self-renewal and differentiation capacity in vivo and competed normally with WT BM in both primary and secondary recipients, suggesting that Smad-mediated BMP signaling is dispensable for HSC function in adult hematopoiesis.
Although the majority of the hematopoietic progenitors screened lacked both Smad1 and Smad5, it could be argued that the lack of phenotype is the result of the very few remaining cells still expressing WT Smad1 or Smad5. By plating sorted donor cells and screening individual colonies for deletion of the floxed Smad genes, we could verify that cells with persistent Smad1 or Smad5 expression were close to absent in BM long-term after transplantation. Furthermore, we could not detect any differences in any hematopoietic parameters analyzed between mice having 100% deletion of both Smad1 and Smad5 and mice with slightly lower deletion efficiencies.
In the light of previous findings, our study suggests that the developmental context is an essential factor in determining how cells respond to growth factors. Several groups have shown the importance of functional BMP signaling for the formation of mesoderm and initiation of hematopoiesis in the developing embryo.7,8,11,12 In addition, Bhatia et al demonstrated that high concentrations of BMP4 maintain proliferation of human cord blood HSCs in vitro.10 Interestingly, the same study demonstrated significant expression of ALK3 and ALK6 in HSCs derived from human cord blood, whereas the expression level of these BMP type 1 receptors in BM derived HSCs was very low to undetectable.10 The absence of these BMP receptors has also been documented in murine BM.22 Intriguingly, we show here that ALK3 is expressed in primitive fetal liver cells, although ALK6 is still undetectable, indicating that this cell population might respond differently to BMP compared with BM cells. Unexpectedly, fetal liver cells deficient in both Smad5 and Smad1/5 were still able to long-term reconstitute lethally irradiated recipients in a multilineage manner. Together, this implies that that ALK6 may be an important type 1 receptor for BMP signaling in hematopoietic cells and that murine HSCs of very early ontogeny might reflect a unique population with a distinct response to growth factors.
We could demonstrate that the related Smad8 is not expressed in BM cells lacking Smad1 and Smad5, suggesting that redundancy between related Smads is not accountable for the lack of phenotype in vivo observed in adult hematopoiesis, despite perturbed BMP signaling. We can, however, not rule out that crosstalk with other pathways might influence the readout of our experiments. The diverging result seen when blocking the entire TGF-β family signaling by overexpressing the inhibitory Smad738 or knocking out the common Smad436 suggests that this might be an option. It has, for example, been shown that crosstalk between BMP and Wnt signaling is vital during early embryonic development in Xenopus.39 In addition, BMP has been reported to activate alternative pathways, such as the mitogen-activated protein kinase cascade in other cell types.40 A possible role for noncanonical Smad-independent BMP signaling in hematopoietic cells might explain the differences between the phenotype obtained in our study and studies where the entire TGF-β signaling pathway was inhibited or components of the BMP signaling pathway upstream of the Smads have been removed. To address this, further investigations are required.
In conclusion, it is well known that several components of the BMP signaling cascade, including Smad5, are important regulators of hematopoiesis during early embryonic development. By investigating the effect of concurrent deletion of Smad1 and Smad5 in both fetal liver and adult hematopoiesis, we have hereby covered the role of canonical BMP signaling throughout ontogeny of murine hematopoiesis. Our results demonstrate that, in contrast to its crucial role during the patterning of hematopoiesis in the embryo, Smad-mediated BMP signaling is dispensable for normal HSC function in both adult and fetal liver hematopoiesis.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Dr Jennifer Moody for helpful advice and discussions, Dr Anita Roberts for kindly sharing the Smad1fl/fl mice, Dr An Zwijsen and Dr Lieve Umans for generously letting us use the Smad5fl/fl mice and for valuable input to this paper, the personnel in the animal facility for taking care of the mice, and Zhi Ma for expert assistance with cell sorting.
This work was supported by the Swedish Cancer Society (S.K.), the European Commission (integrated projects CONCERT, PERSIST, small/intermediate project STEMEXPAND), the Swedish Research Council (project grant, S.K.; Hemato-Linne program project grant, directed by S.K.), the Swedish Children Cancer Foundation (project grant, S.K.; Postdoctoral position grant, G.K.), Lund University Hospital (Clinical Research Award; S.K.), and the European Union (grant T-Angiovasc QLG1-CT-2001-01032; T.G.). The Lund Stem Cell Center has been supported by the Swedish Foundation for Strategic Research (Center of Excellence grant in life sciences, 2002-2009).
Authorship
Contribution: S.S. designed and performed research and wrote the manuscript; G.K. designed experiments, performed laboratory work, analyzed data, and contributed to writing the manuscript; M.E. performed the histopathologic analysis; K.O., P.J., and K.-i.M. performed laboratory work; M.S. and T.G. characterized and contributed with mice vital for this study; and S.K. directed research, designed experiments, and contributed to writing the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
The current affiliation for S.S. is St Vincent's Institute of Medical Research, Fitzroy, Victoria 3065, Australia.
Correspondence: Stefan Karlsson, Molecular Medicine and Gene Therapy, Lund University Hospital, BMCA12, 221 84 Lund, Sweden; e-mail: Stefan.Karlsson@med.lu.se; or Sofie Singbrant, St Vincent's Institute of Medical Research, 9 Princes St, Fitzroy, Victoria 3065, Australia; e-mail: ssingbrant@svi.edu.au.
References
Author notes
S.S. and G.K. contributed equally to this study.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal