Abstract
Abstract 537
Single-arm pilot and Phase II trial data suggested that the combination of fludarabine and alemtuzumab (FluCam) may improve outcome for patients (pts) with relapsed or refractory chronic lymphocytic leukemia (CLL). To validate these observations, a Phase III, multicenter, open-label, randomized study was conducted to compare the efficacy and safety of FluCam vs. fludarabine (Flu) alone as second-line therapy for pts with relapsed or refractory CLL.
Patients with Rai Stages I-IV were randomized to FluCam or Flu using the minimization method to ensure a balance between treatment arms by study center, Rai stage, disease status, age, sex, prior Flu therapy, and maximum lymph node (LN) size. FluCam was administered in Phases A and B. Patients received escalating doses of intravenous (IV) alemtuzumab alone (Phase A). Once alemtuzumab 30 mg IV was tolerated, pts received FluCam as Flu 30 mg/m2 IV followed immediately by alemtuzumab 30 mg IV on days 1-3 of a 28 day cycle (Phase B). In the Flu arm, pts received 25 mg/m2IV on days 1-5 of a 28 day cycle. For both arms, all pts could receive up to six cycles depending on response and toxicity. All pts received prophylaxis with trimethoprim/sulfamethoxazole DS and famciclovir until CD4+ counts were ≥200 cells/μL. The primary endpoint was progression-free survival (PFS). Secondary endpoints were overall response (OR), complete response (CR), overall and 3 year survival, and safety. The primary analysis was based on the independent response review panel's (IRRP) assessment of response and date of progression for each patient. Two interim analyses were prospectively planned and conducted by the data and safety monitoring board (DSMB) with the final analysis planned after a total of 190 events. The 2nd interim analysis included 139 PFS events and met the pre-specified criteria; the DSMB recommended early study termination.
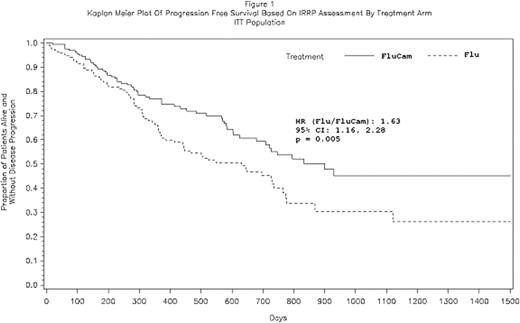
335 pts were randomized (FluCam n=168 and Flu n=167); Rai Stage III-IV: 37%; median age: 60 years; prior Flu therapy: 20% and maximum LN size ≥5 cm: 14%. The median treatment cycles received were 6 for both arms. 60% of FluCam and 64% of Flu pts received 6 cycles of treatment. The median IRRP determined PFS for FluCam was significantly prolonged compared to Flu (29.6 months vs. 20.7 months, respectively; p=0.005; HR 1.63 [95% CI: 1.16, 2.28]; Figure 1). Median PFS by Rai Stage was: Stage I-II - 27.4 months for FluCam (n = 105) vs. 21.3 months for Flu (n = 103), p=0.215; Stage III-IV - 26.1 months for FluCam (n = 61) vs. 12.1 months for Flu (n = 62), p=0.003. Per investigator response assessment, FluCam resulted in significantly higher OR and CR rates (OR: FluCam 84.8% vs. Flu 67.9%, p<0.001; and CR: FluCam 30.4% vs. Flu 16.4%, p=0.002). The IRRP assessment of response was not completed for all pts and is not availabel for the 2nd interim analysis. No differences in survival have been observed (FluCam 37 deaths and Flu 41 deaths) with a median follow up of 17 months. Adverse events (AEs) occurring in >10% of the pts included pyrexia, neutropenia, leukopenia, thrombocytopenia, anemia, chills, lymphopenia, rash, infusion related reactions, nausea and urticaria in the FluCam arm; and, neutropenia, thrombocytopenia, anemia and leukopenia in the Flu arm. Treatment-emergent grade 3/4 thrombocytopenia (18% vs. 22%), neutropenia (60% vs. 66%) and anemia (13% vs. 22%) were comparable in FluCam vs. Flu arms. Overall, 33% (n=54) of pts in the FluCam arm experienced a SAE vs. 26% (n=42) in the Flu arm. Reported SAEs for neutropenia were 4.9% in the FluCam arm and 1.8% in the Flu arm; however, febrile neutropenia was similarly reported in the two arms 3.7% vs 3.6% of pts, respectively. Infections including CMV occurred in 47% and 35% of the FluCam and Flu pts, respectively. Symptomatic CMV infection occurred only in the FluCam arm in 8% of pts, of which 1% were SAEs and 0% classified as grade 4 or higher. Deaths occurring on therapy or within 30 days after last dose were 2% on the FluCam arm vs. 5% on the Flu arm.
The 2ndinterim analysis indicates that the combination of FluCam is superior to Flu as second-line therapy for pts with relapsed or refractory CLL, including those with advanced disease stage. With significantly longer PFS, higher OR and CR rates, an acceptable safety profile and a convenient administration regimen, FluCam may be an additional second-line treatment option for pts with relapsed or refractory CLL.
Engert:Genzyme Corporation: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Off Label Use: Alemtuzumab (Campath, MabCampath) is indicated for the treatment of CLL. This trial examined the use of alemtuzumab in combination with fludarabine monophospate.. Gercheva:Genzyme Corporation: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Robak:Genzyme Corporation: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Galina:Genzyme Corporation: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Wu:Genzyme Corporation: Employment. Sirard:Genzyme Corporation: Employment. Elter:Genzyme Corporation: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal