Abstract
Abstract 2281
Poster Board II-258
A decision analysis using the Markov process is a flexible and convenient analytical method that tracks the clinical events that occur after a certain decision with different probabilities and utilities over time. To address the role of allogeneic hematopoietic cell transplantation (allo-HCT) for acute myeloid leukemia (AML) in CR1, we performed a Markov decision analysis using newly collected clinical data from 2029 patients.
Probabilities and other outcome data were derived from a database of adult AML patients that was constructed from case report files collected for this study. We included patients who were diagnosed with AML other than M3 between 1999 and 2006, aged 16 to 70 years, and who had achieved CR1 after 1 or 2 courses of induction chemotherapy. Using the software package TreeAge Pro 2009, we constructed a Markov decision model that compared 2 strategies: allo-HCT in CR1 (HCT group) and no allo-HCT in CR1 (CTx group). Possible health states considered in each decision included, for the HCT group, 1) no relapse without chronic GVHD, 2) no relapse with chronic GVHD, 3) relapse, and 4) dead, and, for the CTx group, 1) no relapse, 2) relapse, 3) second remission, 4) after salvage allo-HCT, and 5) dead. Quality-of-life (QOL) adjustments were made by incorporating time trade-off utilities that were derived from a questionnaire to 12 physicians who were familiar with the treatment of AML. The cycle length between state transitions was set at 3 months.
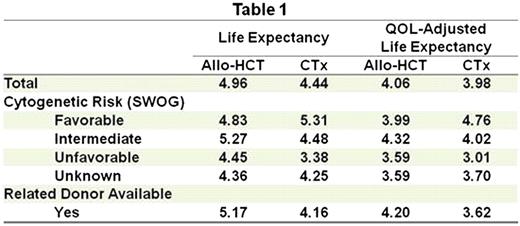
A total of 2029 patients were eligible for this analysis. The median age was 50 years, and the median follow-up of the surviving patients was 4.10 years. The proportions of patients with favorable, intermediate, unfavorable and unknown cytogenetic risk by SWOG criteria were 20%, 54%, 17% and 9%, respectively. Therapies performed at CR1 were allo-HCT in 494 patients (24%) and chemotherapy in 1535 patients (76%). Among 1076 patients whose HLA typing was performed for allo-HCT in CR1, 431 had HLA-matched or 1-antigen-mismatched related donors. Life expectancy and quality-adjusted life expectancy for the HCT and CTx groups in each risk category are summarized in Table 1. Life expectancy of the HCT group was longer than that of the CTx group (4.96 years vs. 4.44 years). However, quality-adjusted life expectancy of the HCT group was comparable to that of the CTx group (4.06 years vs. 3.98 years) due to a larger reduction of expected life length in the HCT group after QOL adjustment. In a subset analysis of the CTx group, patients with more favorable cytogenetic risk had a longer life expectancy. Whereas allo-HCT in CR1 was associated with a shorter life expectancy in patients with favorable-risk AML, allo-HCT in CR1 was associated with a longer life expectancy in those with intermediate-risk or unfavorable-risk AML. Adjustment for QOL did not change the preferred decision in the intermediate- and unfavorable-risk groups, although the survival advantages for allo-HCT in CR1 were less than those without QOL adjustment. In a subset of patients who had related donors, both life expectancy and quality-adjusted life expectancy were longer in the HCT group.
The results of our decision analysis using the Markov process indicated that patients with intermediate- or unfavorable-risk AML have a longer life expectancy and quality-adjusted life expectancy with a decision of allo-HCT in CR1. Our results also showed that a Markov decision analysis that incorporates QOL may be useful as a decision-making tool for patients who might be candidates for allo-HCT.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal