Abstract
INTRODUCTION: Impaired renal function in patients with multiple myeloma (MM) is associated with adverse clinical outcome. Historically, the renal status is reported as serum creatinine (SCr) only. Introduction of novel agents (thalidomide, lenalidomide, bortezomib and pegylated liposomal doxorubicin) in MM therapeutics has improved clinical responses and overall survival. An important question that remains largely unanswered is if these agents deliver equal benefit to patients with impaired renal function. Thus we investigate the overall benefit of novel agents in MM patients with renal impairment.
METHODS: All MM patients treated between January 2000 and December 2007 at Roswell Park Cancer Institute (RPCI) where included in this analysis. Extent of disease was assessed based on the Durie-Salmon staging system. We determined glomerular filtration rate (GFR) to asses renal function more accurately. GFR was calculated utilizing the Modification of Diet in Renal Disease (MDRD) equation. Patient cohorts were defined based on severity of renal impairment per the National Kidney Foundation-Kidney Disease Outcomes Quality Initiative (NKD-KDOQI) guidelines based on the GFR value into normal >90, mild 60–89, moderate 30–59, severe 15–29 and kidney failure <15 ml/min/1.73m^2. Because there were only few patients in the last two groups (GFR of 15–29 =11 and GFR <15 =13), they were combined into a single cohort of severe renal impairment (GFR <30=24). We also defined patient cohorts based on a previously reported classification in MM wherein patients were divided based on severity of renal impairment into normal >80, mild 50–80, moderate 30–49 and severe <30 ml/min/1.73m^2. Survival curves and median survival were calculated by Kaplan-Meier method. Survival differences were calculated using a log-rank test. A 0.05 nominal significance level was used in all statistical testing.
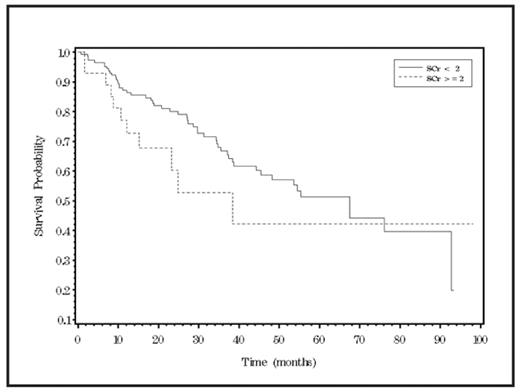
RESULTS: A total of 175 patients (M=88, F=87) with a median age 60 years (range 34–88) were evaluated. Majority of patients had stage III disease (64%), 57.1% had IgG myeloma and lytic bone disease present in 67.4% of the patients. Median values for SCr, serum calcium, blood urea nitrogen, serum albumin and hemoglobin were 1.1mg/dl (range-0.5–10.3), 9.5 mg/dl (range-7.5- 7.2), 18 mg/dl (range-5–107), 4.0 g/dl (range-2.2–5.5) and 11.5 g/dl (range-6.8–17.1) respectively. SCr >2 mg/dl was seen in only 16% patients and 41.1% patients had GFR in the range of 60–89 ml/min/1.73m^2. Patients received an average of 2 treatments. Use of at least one of the novel agent was seen in 86.3% patients (Immunomodulatory drugs =130, Bortezomib = 97, Pegylated liposomal doxorubicin =65). Median survival for patients with SCr <2.0 and SCr>2.0 was 67.4 months (45.4 – 92.7 months) and 38.4 months (15.3 months – not reached) respectively. Median survival for patients with normal GFR and mild, moderate and severe renal impairment based on the NKD-KDOQI guidelines was 76.1 months (37.5 months – not reached), 55.32 months (34.8 months – not reached), 67.4 months (45.4 months – not reached) and 24.9 months (15.3 months – not reached) respectively. Median survival for patient cohorts defined based on a previously reported classification in MM was similar to that seen with the NKD-KDOQI classification. We noted no significant survival advantage of normal renal function over renally impaired patients (Figure 1 & 2) despite evaluations based either by using the traditional approach of SCr (p=0.15; log rank test) or by more sensitive renal function assessment by GFR (p=0.21; log rank test).
CONCLUSIONS: We conclude that in MM patients the previously reported and commonly perceived adverse prognosis imparted by impaired renal function can no longer be validated and that routine incorporation of novel agents has overcome this adverse prognosis. These findings are consistent with the impact of novel agents reported in context with MM with aggressive molecular profiles.
Kaplan-Meier curve for survival by serum creatinine (SCr)
Kaplan-Meier curve for survival by serum creatinine (SCr)
Disclosures: Miller:Celgene: Consultancy, Honoraria, Speakers Bureau; Millenium: Consultancy, Honoraria, Speakers Bureau. Chanan-Khan:Celgene: Consultancy, Honoraria, Speakers Bureau; Millenium: Consultancy, Honoraria, Speakers Bureau.
Author notes
Corresponding author

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal