Abstract
Background: Factors influencing participation in clinical trials have been reported, but few studies analyze the combination of issues which affect overall patient accrual. When designing clinical trials, investigators are aware that some potential subjects may not be candidates for research participation and other subjects will decline. Total sample size is further affected by protocol-defined eligibility. BABY HUG (NCT00006400) is a randomized, double-blind, placebo-controlled Phase III clinical trial of hydroxyurea (HU), which had an enrollment goal of 200 infants, age 9–17 mo, with Hb SS or Sβ0-thalassemia. A number of screening tests (spleen scan, 99mTc-DTPA renal clearance, abdominal sonogram, transcranial Doppler ultrasound, neuropsychological testing, blood analyses) were required prior to study entry and at exit, with at least monthly follow-up visits during the 2-year study period. To enhance recruitment, an anonymized registry of potential subjects was created to identify factors affecting enrollment.
Methods: BABY HUG study coordinators considered all infants with an FS/SS diagnosis on newborn screening who were less than age 17 mo. The coordinators developed an IRB-approved spreadsheet to record the number of potentially eligible subjects; whether parents were approached and, if not, why not; and reasons of those approached for participating or declining. Most of these reasons could be categorized into one of several common themes.
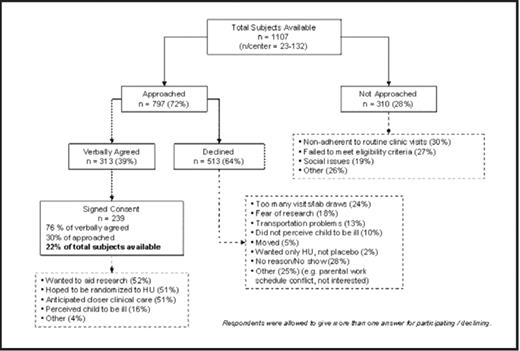
Results: Of 1107 potential participants (23–132/center) identified between October 2003 and June 2007, 239 (22%) entered the screening process and 193 (17%) were randomized. More than 25% were not approached for various reasons, including poor adherence to standard clinical care or failing to meet eligibility criteria. Factors influencing enrollment decisions are shown in the figure. The willingness to contribute to medical knowledge, the hope of being randomized to receive the investigational drug (HU), and the desire for closer clinical care were the most common reasons for participating in BABY HUG. Conversely, fear of research, transportation problems, parental work schedules and the demanding nature of the study (with frequent blood samples and clinic visits) were the primary reasons for parents declining. Disease severity had less impact on decision-making, perhaps reflecting the often asymptomatic history of potential subjects.
Conclusion: The time and effort required by multiple screening tests and frequent visits impact the willingness of eligible subjects to participate in a study. Similar trials may require a larger pool of potential participants than expected to meet accrual goals. Factors that influence subject availability and the decision-making process of participating families must be considered for successful recruitment.
Disclosures: No relevant conflicts of interest to declare.
Author notes
Corresponding author

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal