Clinical proteomics has been applied to define a specific polypeptide profile for acute graft-versus-host disease (aGvHD) after allogeneic hematopoietic stem cell transplantation (allo-HSCT). This profile can then be used for accurate prediction of the occurrence and recurrence of aGvHD.
A biomarker has been recently defined by the Biomarkers Definitions Working Group as “a characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathological processes, or pharmacological responses to a therapeutic intervention.”1, pg91 Biomarkers, therefore, have potential values in monitoring health status. They can be used as a diagnostic tool for disease detection and staging, as well as a tool for disease prognosis and prediction of therapeutic outcome or response to an intervention. Additionally, the ideal biomarkers should also be able to predict recurrence or relapse of the disease after treatment.
After the completion of the Human Genome Project, the post-genomic research has been developing rapidly and has focused largely on translating the genomic information to clinical applications. Clinical proteomics has become one of the most interesting fields with an ultimate goal of biomarker discovery for earlier and more accurate diagnosis, and for more accurate prognosis of the disease. Although the main focuses are diagnostics and biomarker discovery, clinical proteomics also covers identification of new therapeutic targets, drugs, and vaccines for better therapeutic outcomes and successful disease prevention. Recently, a group of proteomists, clinicians, biochemists, chemists, bioinformaticians, and statisticians from more than 25 institutions worldwide have discussed and begun to define the field and to set adequate standards for clinical proteomics.2 Through this collaborative effort, clinical proteomics has been defined as “the application of proteomic analysis with the aim of solving a specific clinical problem within the context of a clinical study.”2,pg149 A clinical proteomic study should begin with a well-framed clinical question or problem, followed by selection of the appropriate study populations, samples to be analyzed, and technology to analyze the samples.2
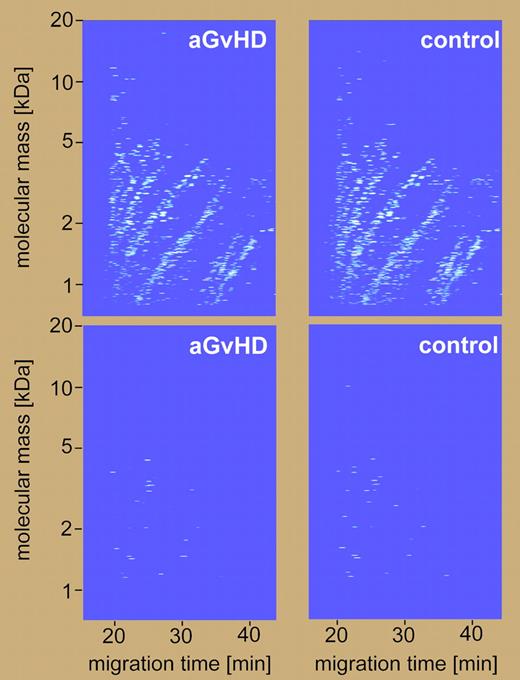
Analysis of urinary polypeptide patterns of aGvHD and non-aGvHD using CE-MS. See the complete figure in the article beginning on page5511.
Analysis of urinary polypeptide patterns of aGvHD and non-aGvHD using CE-MS. See the complete figure in the article beginning on page5511.
Although some single biomarkers have been identified for particular diseases, it has been suggested that a single ideal biomarker may not exist for every disease. Evaluation of a panel of multiple potential disease-specific biomarkers is, therefore, crucially required; proteomics serves as an important tool for such purpose to examine the molecular signature of proteins in a biological sample (proteome profiling). One of the proteomic methodologies that is suitable for proteome profiling is capillary electrophoresis coupled to mass spectrometry (CE-MS).3 Advantages of CE-MS include automation, high-throughput capability, high sensitivity, and less demand of sample volume.
In this issue of Blood, Weissinger and colleagues have applied state-of-the-art proteomic technology using CE-MS to evaluate urinary polypeptide profiles (patterns) of 141 allo-HSCT patients from 5 centers in Germany and the United States. Using this technique for analyzing a training set of 13 urine samples from patients with aGvHD and 50 samples from patients without aGvHD, they defined the aGvHD-specific urinary polypeptide pattern. A model of multiple potential biomarkers containing 31 polypeptides allowed accurate classification of urine samples in the training set with a sensitivity of 100% and specificity of 98%. This panel of multiple potential biomarkers was then used for classifying 599 urine samples in the validation set and provided satisfactory sensitivity and specificity (83.1% and 75.6%, respectively). In addition, urine samples from healthy individuals and patients with other diseases were also analyzed as the quality-control set. Moreover, their findings, particularly the classification factor or support vector machine (SVM) score, could be used for prediction of the occurrence and recurrence of GvHD.
This study is an excellent model to underscore the applicability of clinical proteomics to diagnostics and prognostics. However, a limitation of CE-MS should be also noted. Any proteins with molecular masses greater than 20 kDa are not suitable for CE-MS analysis and, thus, require other complementary methods.
Conflict-of-interest disclosure: The author declares no competing financial interests. ■

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal