Abstract
Introduction: In Japan, myelodysplastic syndromes (MDS) and aplastic anemia (AA) are the most common transfusion-dependent anemias. Deferasirox (Exjade®, ICL670) is a once-daily, oral iron chelator for the treatment of transfusional iron overload; its efficacy and tolerability have been established in adults and children with a range of transfusion-dependent anemias, including MDS. Here, deferasirox has been evaluated in Japanese patients and the pharmacokinetics (PK) and pharmacodynamics (PD) compared with those seen in a US study.
Methods: The tolerability, PK and PD of deferasirox have been assessed in a Phase I, open-label, unblinded, dose-escalation trial (1101) in Japanese patients with MDS and AA in 9 centers. Deferasirox was administered as a single dose of 5 (n=6), 10 (n=7), 20 (n=6) or 30 mg/kg (n=7). After a 7-day break, daily doses were administered (same doses/same patients) for 7 consecutive days. Data were compared with a study (0104) in 24 Caucasian β-thalassemia patients (age range 18–39 years) who were administered doses of deferasirox of 10, 20 or 40 mg/kg/day for 12 days. Throughout the study, iron intake and excretion were rigorously measured.
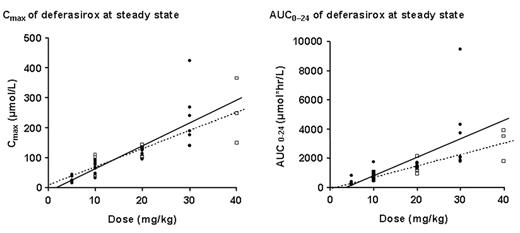
Results: A total of 26 Japanese patients (8 male, 18 female; mean age 65.6 years; 8 aged <65 years) completed single- and multiple-dosing periods (16 MDS; 6 AA; 4 other anemias). After a single dose and also at steady state, linear PK (Cmax and AUC) were observed at all doses. The figures show Cmax and AUC at steady state for both the Japanese patients (closed circles, solid lines) and the Caucasian patients (open squares, dotted lines), and demonstrate that PK were similar.
Dose-dependent iron excretion (range 0.07–0.61 mg iron/kg/day) was observed, similar to that in the Caucasian patients (range 0.12–0.45 mg iron/kg/day). A linear relationship (PK/PD) was noted between AUC and iron excretion. Deferasirox was well tolerated, with generally infrequent and mild adverse events (AEs). The most common AEs related to deferasirox were diarrhea (n=2 each after single and multiple doses of 30 mg/kg/day) and nausea (n=1 each after multiple doses at 10 and 30 mg/kg/day). One patient had two serious AEs, pyrexia and duodenal ulcer, both suspected to be related to study drug, after multiple doses of 30 mg/kg/day.
Conclusions: Deferasirox is well tolerated both after single and multiple doses in Japanese MDS/AA patients, with similar PK/PD parameters to those in the Caucasian β-thalassemia cohort. Exposure to deferasirox in the Japanese patient cohort was dose dependent with a linear PK/PD relationship resulting in dose-dependent iron excretion. These data suggest that deferasirox is effective and well tolerated regardless of underlying disease, race or age.
Disclosures: H Tanii, M Kondo, J Taniguchi, H Schran and B Rabault are Novartis employees.
Author notes
Corresponding author

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal